Each summer, the University of Rhode Island Graduate School of Oceanography (GSO) hosts undergraduate students from all over the country to participate in oceanographic research. These Summer Undergraduate Research Fellows (SURFOs) have not only been working with GSO scientists, but they have spent part of their time learning how to communicate this science to the public. Read on to find out what they have been up to, and why they everyone should be as excited as they are about their work.
Authors: Allyson Murray, Ellie Tan, Samantha Vaverka, Joseph Barnes, and Gibson Leavitt

[hr][divider][/hr]
Using sediment to understand Haiti’s climate and tectonic history
Allyson Murray – Stockton University
Advisor- Dr. Milene Cormier
Earthquakes are everyday hazards which can sometimes cause severe destruction. On January 12, 2010 a magnitude 7.0 earthquake struck Haiti, and severely damaged the capital city of Port-au-Prince and the surrounding areas, causing casualties and economic losses. The 2010 earthquake exposed the limited knowledge of the local tectonics, especially the Enriquillo-Plantain Garden Fault (EPGF). The EPGF marks a major transform boundary between the Caribbeanand North American plates, similar in it activity to the San Andreas fault in California. Meaning that it is an area where earthquakes are likely to occur. The EPGF bisects the southern part of Haiti, passes through Port-au-Prince, before potentially plunging below Lake Azuei. Lake Azuei is a large (6.2 miles x 15.5 miles), brackish lake located in Haiti about 18.6 miles east of the capital city of Port-au-Prince.
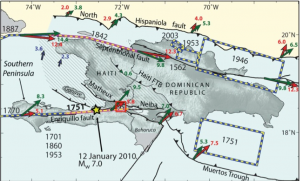
Each local earthquake disrupts the sedimentary layers accumulating at the lake floor. Over geological time, successive earthquakes result in large-scale deformationof these sediment layers into features such as faults, fold, and landslides. My project aims to image such seismotectonic deformation recorded within the lake sediments, and by dating some of the sediments, to quantify the rate at which deformation occurs. This information will help identify those regions at greatest risk in the next earthquake. When communicated to local and national governments, it can be applied to reinforce infrastructure where needed, educate the public, and prepare the emergency response for the upcoming disaster.
For my project this summer, I am analyzing data retrieved during a 2017 research expedition to lake Azuei funded by the National Science Foundation.

The main objective of that expedition was to image the geological structures hidden beneath the surface of the lake bed, which was accomplished by using seismic reflection methods. Seismic reflection is a geophysical method which involves generating a sound wave and recording the time it takes for the sound wave to reflect off the interfaces between geological layers and reach the receiver at the surface. The seismic source and the hydrophones that record the reflected signal are towed behind a boat while traveling at constant speed. This allows for geological features beneath the lake bed to be imaged as vertical profiles. This expedition also collected two sediment cores from the lake bed. The cores consist of cylindrical samples of sediments a few inches in diameter. They sampled a few feet of sediments beneath the lake floor.

The high-resolution seismic reflection profiles reveal an ancient shoreline submerged ~11 m below the present lake level. Indeed, shorelines tend to be marked by a “shoreline angle”, where gentler slopes underwater meet steeper slopes on land. These breaks in slope can be identified on high-resolution seismic profiles and, if enough seismic profiles are available, they can be accurately mapped in 3D. These shorelines are useful markers of original horizontality. If the shoreline is not horizontal, as in Lake Azuei, it quantifies relative vertical deformation and highlight which structures have been active since the shoreline became submerged. Additionally, if paleoshoreline can be dated, they document the rate of relative vertical motion. Rate of deformation is important information to assess seismic risk. The faster the rate, the higher the risk; i.e. the sooner the next earthquake is likely to occur.
We propose that this paleoshoreline at ~11 m depth corresponds to a known period of maximum aridity dated around ~1,000 years before present and recorded throughout the Caribbean, where it is referred to as the “Terminal Classic Drought”. In order to test our hypothesis, we are currently analyzing our sediment cores for Carbon-14 datable material, such as shells or tree bark. Carbon-14 is a dating technique, which allows for the age organic material to be dated based on the amount of Carbon-14 left in the fossil after death. This is a fairly reliable dating method for matter younger than 50,000 years.
The sediment cores taken from the lake bed sample layers identifiable in the seismic reflection profiles taken. One of the layers sampled extend laterally below the paleoshoreline, which implies that layer is older than the paleoshoreline. This would mean the age found at the bottom of the sediment core, by dating with Carbon-14, will predate the shoreline. This will give an approximate age range for the shoreline and provide new additional insight into the local climate history, especially in the support or rejection to the theory that a period of maximum aridity affected the entire Caribbean region.
Another important aspect of my project is to analyze the seismic reflection profiles in order to find structures that reflect seismic activity. The fault that potentially runs through the southern portion of Lake Azuei causes deformation in the lake bed. The lake bed is generally flat, which suggest it is infilled with turbidites. Turbidites are sedimentary layers that form when sand and mud resting loosley on a slope are set in motion by events such as earthquakes or storm waves. The layers are coarse at the bottom then get finer in size at the top. Turbidites are originally deposited horizontally in the deepest areas, so imaging the deformation of turbiditic layers will provide insight into seismic deformation.
This project is carried out in direct collaboration with Haitian scientists at the State University of Haiti and at the Haitian Office of Mining and Energy (BME). More information about the Lake Azuei project can be found on www.projectlakeazuei.org
[hr][divider][/hr]
Building LoBSTAS: Eyes on the Seafloor
Elizabeth Tan – Wheaton College (IL)
SURFO advisor: JP Walsh and Brice Loose
Did you know that fish and other marine organisms can suffocate underwater? When the dissolved oxygen concentration in water drops below a certain concentration in a phenomenon known as hypoxia, marine organisms become stressed as they are lacking oxygen. In extreme cases (anoxia), most of them die! While such tragic events are generally called fish kills, the casualties can also include other marine creatures like crabs, shrimps, and eels.

In hypoxic or anoxic regions (typically along coastlines), local economies can be greatly affected. Shellfishery yield and fishing catches decline. Houses along the coastline drop in value. Once popular beaches become deserted, thanks to dead crabs strewn everywhere and smelly, greenish seawater. These are extreme cases from fish kill events; essentially, floating fish means a sinking economy.
So what drives hypoxia?
There are many processes that can cause low dissolved oxygen levels in the water, but I group them into two main ideas: processes that (1) use up oxygen with respiration, and processes that (2) help replenish oxygen but may be too slow or ineffective. Nutrient loadingcauses algal blooms; when these blooms die, this can cause hypoxic ‘dead zones’ on the seafloor due to lots of decaying organic matter taking oxygen out of the water column during decomposition processes. To replenish dissolved oxygen in these dead zones, vertical and horizontal water circulation is critical. In coastal areas where there is a layer of warm fresh water above cooler salt water, the highly stratified layers do not mix easily without the help of waves, wind-driven currents, and tidal currents.
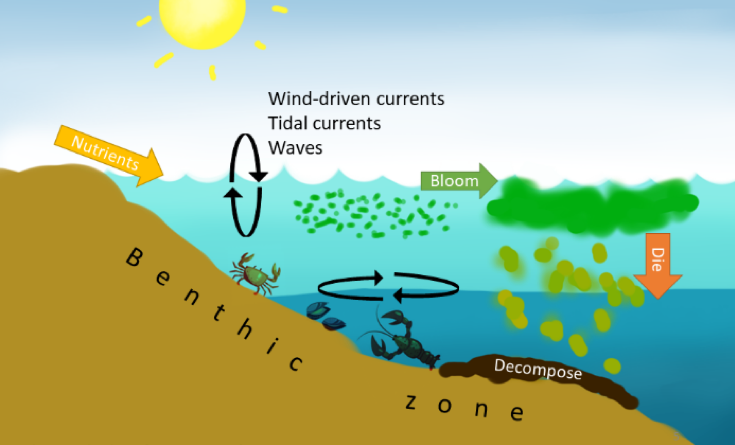
Benthic organism icons from www.123rf.com
So, if you’re a slow-moving benthic dweller on the seafloor caught in one of these dead zones, you’re in big trouble. The available, breathable oxygen around you is dropping dangerously low, and you can’t move out of the growing dead zone fast enough! Basically, the benthic zone is most susceptible to hypoxia.
My project: Low-cost Benthic Sensing Trap-Attached System (LoBSTAS)*
Although many driving processes of hypoxia are well studied, the interaction between benthic physical processes, benthic organisms, and hypoxia is poorly understood. Thus, I have been working on a summer research project to design and build an autonomous benthic weather station to monitor seabed dynamics. An onboard camera will record time-lapse imagery and video segments to reveal seafloor currents, benthic storms, and sediment evolution in time. Would we see less water circulation in dead zones? Would a benthic storm re-oxygenate a dead zone? Would we see much more benthic organism activities in water with strong circulation since there is now much more dissolved oxygen at the bottom? With data from the LoBSTAS, we will be able to investigate these questions.


Since an important requirement is that the unit is low-cost, each unit is made with off-the-shelf hardware and open-source software such as Linux and Python (Figure 2). With costs many times cheaper than industrial underwater research systems, these LoBSTAS can be easily reproduced for mass deployment to acquire an extensive set of data. The low cost and reproducibility will also make the LoBSTAS an effective tool for researchers, managers, or the public with limited finances.

To deploy the BROS, I will first configure the mission settings from a phone or laptop, then attach units to lobster cages (or simply tape them to rocks and attach a small rope to it) and drop them to the seafloor. After 3-5 days, I will retrieve the unit to copy out the pictures and videos for analysis. From images of the benthic environment, we can quantify characteristics such as sediment type, vegetation cover and species, benthic organism presence, and water turbidity. In addition, videos help us understand local water dynamics through the velocity of particles and seaweed motion. All these characteristics vary from location to location, but we hypothesize that hypoxic regions with low dissolved oxygen should have slower water circulation, lesser benthic fauna and flora, and sediment with more (decomposing) organic matter.

I am currently working on a prototype #3 to make night imaging possible. This would reveal day and night patterns in dissolved oxygen levels due to photosynthesis and investigate the higher rick of hypoxia during the night. I am also preparing for a full deployment to capture imagery and dissolved oxygen levels at Wickford Harbor. As the BROS continue to keep an eye on benthic environments, I hope that the combined visual and numerical data will help us better understand the link between benthic processes and the variability of dissolved oxygen in space and time. With a better grasp on the driving processes of hypoxia, we will be able to care for our coastal areas and prevent the occurrence of fish kills due to hypoxia.
*Want to build a LoBSTAS? Building recipe will be available at geotracerkitchen.org
http://https://youtu.be/OT_flC3ZvS4
Video caption: Fast currents at the seafloor (16ft depth) near the GSO dock. There was an unusually high number of big and small fish near the seafloor!
Standalone link: https://youtu.be/OT_flC3ZvS4
[hr][divider][/hr]
Tiny Plants of the Sea: Helpful Friend or Toxic Foe?
Samantha Vaverka – Augustana University
SURFO Advisor: Dr. Bethany Jenkins
What if I told you that around 70% of the world’s oxygen comes from a source invisible to the naked eye? Years ago, I would never have believed such a crazy idea. Growing up in the Midwest, I never expected to fully grasp the diversity of the ocean. Little did I know I was completely unaware of one of the most crucial parts: tiny plants of the sea, otherwise known as phytoplankton. While these “micro-plants” operate in a similar manner to land plants, they differ in a few key ways. One of these is their seasonal change in abundance, increasing up to counts of hundreds of thousands of cells in each liter of ocean water. While this may seem insane, it makes sense that it takes so many tiny plants to provide such a high level of oxygen. It turns out that phytoplankton do much more for us than their small size would suggest.
 Now imagine if these helpful phytoplankton friends began to produce a toxin harmful to humans. With such high numbers, the toxin could spread throughout the waters, passing through organisms that filter water to obtain food. This scenario happens to be the case for one type of phytoplankton in Narragansett Bay, RI. Organisms in the Bay of the genus Pseudo-nitzschia have recently begun to produce a toxin called domoic acid. Currently it is not known whether there is a new species present, or if some environmental factor is causing an already present species to produce the toxin. While the toxin levels necessary to cause effects in humans are high, the toxin can accumulate in shellfish, causing an illness known as amnesic shellfish poisoning in humans upon consumption. As a result, this topic has become of great interest to researchers studying the ecology of Narragansett Bay. Although the shellfish are tested, protected, and safe to eat as there has not been a poisoning event in RI to date, the science behind such an event is fascinating. So what is causing this seemingly harmless organism to produce such a potent toxin?
Now imagine if these helpful phytoplankton friends began to produce a toxin harmful to humans. With such high numbers, the toxin could spread throughout the waters, passing through organisms that filter water to obtain food. This scenario happens to be the case for one type of phytoplankton in Narragansett Bay, RI. Organisms in the Bay of the genus Pseudo-nitzschia have recently begun to produce a toxin called domoic acid. Currently it is not known whether there is a new species present, or if some environmental factor is causing an already present species to produce the toxin. While the toxin levels necessary to cause effects in humans are high, the toxin can accumulate in shellfish, causing an illness known as amnesic shellfish poisoning in humans upon consumption. As a result, this topic has become of great interest to researchers studying the ecology of Narragansett Bay. Although the shellfish are tested, protected, and safe to eat as there has not been a poisoning event in RI to date, the science behind such an event is fascinating. So what is causing this seemingly harmless organism to produce such a potent toxin?
Bacteria on Phytoplankton
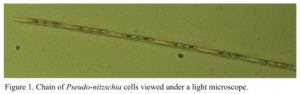
While the reasons behind domoic acid production by certain Pseudo-nitzschia species remain unclear, a few patterns have been noted. One of these patterns has become the central focus of my SURFO project for the summer. Interestingly, these tiny marine plants are often associated with bacteria, as seen by the arrow in Figure 2.
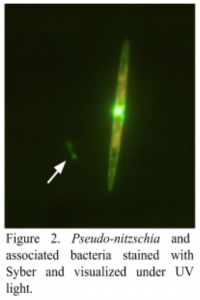
In fact, the bacteria can even attach to the surface of the organisms, forming a miniature bacteria community, also known as a microbiome. Just as you and I have friends that we hang out with on a regular basis, so do the Pseudo-nitzschia cells. In other words, they are most likely to be found with certain bacteria attached to them. Although we do not know which specific bacteria these are, we do know that they can perpetuate or even increase the production of domoic acid. As a result, my research involves identifying the specific species of Pseudo-nitzschia responsible for the toxin production in the Bay, as well as the bacteria associated with these tiny plants of the sea.
Identifying Associated Bacteria
In order to study the bacteria that may be involved in domoic acid production, tools of molecular biology were used. In layman’s terms, this primarily involved looking at the DNA of the bacteria to determine which specific bacteria friends were present. Water was collected from various sites throughout both upper and lower Narragansett Bay, and filtered to collect the Pseudo-nitzschia cells and associated domoic acid concentrations.

After this, the cells were broken open and the DNA was pulled out, a process called DNA extraction. Specific primers were then used to amplify the highly variable V4 region of the 18S rDNA gene that all phytoplankton have. Also in this process, bacterial DNA was extracted and the 16S gene was amplified. In the near future, thousands of these phytoplankton and bacteria sequences present in our samples will be identified using high throughput, or next generation sequencing technology. What does that all mean? Essentially, we took the bacteria attached to the tiny plant cells, pulled out the DNA of both the plant cells and the bacteria, and made a bunch of copies of a specific part of the DNA which is different in each species of phytoplankton or bacteria. From this, we can tell which of our bacteria friends are present in each different water sample. If we can identify which bacteria are associated with which species of Pseudo-nitzschia, that could provide us with a great deal of information regarding the connections behind toxin production and bacterial involvement.
Why Does This Matter?
Exploring the driving factors involved in domoic acid production by Pseudo-nitzschia will give a better understanding of how and when toxin production will increase and decrease in Narragansett Bay. This will in turn provide for better methods of testing for toxin production, and possible predictors for future toxic events. In a sense, it will be much easier to know when and why the shellfish harvested in the Bay are or are not safe to consume. This directly translates to greater implications in the way of community health, and a greater understanding of the molecular ecology within Narragansett Bay, RI.
[hr][divider][/hr]
Hide-and-Seek with Toxic Phytoplankton
Joseph Barnes, SURFO Student Liaison
SURFO Advisor: Dr. Lucie Maranda
It’s a beautiful sunny summer day. The wind has been steady all afternoon, the waves are calm and tranquil, and the squawking seagulls even manage to seem endearing today. It’s perfect for that clam bake you’ve been planning. On the way home from work, you stop by the local clam shack to pick up the day’s harvest. The shoreline behind the shack is empty, where usually you see the workers harvesting the shellfish, picking off plants and algae from the cages, or maybe even sneaking a few for a late snack. The shack, too, seems oddly devoid of customers. The window is closed, there’s no one around, and suddenly the squawking seagulls seem ominous amid the white noise of an ever-approaching high tide. There is a small sign posted that talks about a sudden increase in microscopic organisms that have started producing acid in the water. The shellfish have eaten them and are now unsafe for humans to eat. The wind blows, crows appear and start cawing, a frantic biker whips past, spreading tumbleweeds that roll and stop at your feet. The perfect day now seems horribly bleak.
That post-apocalyptic analogy may be exaggerating things a bit, but when microorganisms rapidly become abundant and start producing acid in the ocean, things get bad. That’s exactly what Rhode Island’s Narragansett Bay faces each summer as a certain microorganism known as Pseudo-nitzschiareproduces and its population grows exponentially.Pseudo-nitzschiais a type of diatom, a single-celled plant with an outer casing made of silica. It drifts around with the waves, congregating near the surface to receive sunlight so it can photosynthesize to make its own food. Many species of this diatom have lived and thrived in Narragansett Bay together for years, but only recently have scientists documented it producing domoic acid. Domoic acid is a neurotoxin, but as of yet no one knows why these tiny plants produce it.
In Dr. Lucie Maranda’s lab at the University of Rhode Island Graduate School of Oceanography, we are studying the prevalence of Pseudo-nitzschia species in the Bay, tracking their movements to find out if the rapid increase of populations, known as a harmful algal blooms (HAB), is a result of a few cells lying dormant in the Bay and “waking up” under the right conditions, or if external factors from Rhode Island Sound like seasonal tidal cycles, nutrient availability, or even a new Pseudo-nitzschia species, are causing a the HABs.

Seven stations in Narragansett Bay are sampled every other week from one of URI’s small research vessels. Remember to take motion sickness medicine that morning, because the voyage starts at 6 A.M. and treks down to the Atlantic Ocean where the waves are bigger. At each station, a CTD is deployed, which measures water parameters like temperature, salinity, depth, and chlorophyll levels. The chlorophyll levels indicate phytoplankton density, potentially Pseudo-nitzschia. Water samples are taken at the bottom depth, at the chlorophyll maximum- where there is the most phytoplankton- and at the surface. The samples are filtered in a way that all the large and miniscule organisms are filtered out, leaving just organisms around the size of Pseudo-nitzschia. At the same depths, more water samples are taken to be analyzed for nutrient availability, like nitrogen, phosphorus, and silica, which are all elements found naturally in the water that diatoms depend on. The station closest to the ocean serves as our sentinel station, where we can use data from there as a piece of the puzzle in figuring out the progression of domoic acid-producing diatoms into the Bay.

So long as the waves are calm, the sun is shining, and the medication is working, the boat rides are enjoyable. Preferable, however, is the onshore data collection. Integrated vertical phytoplankton tows, with a net that captures phytoplankton no matter their position in the water column, are taken biweekly from two locations covering the East and West passages of Narragansett Bay to further track Pseudo-nitzschiapopulation movement. These samples are also filtered in a way that leaves cells of a desired size. Half the sample is preserved to count the number of Pseudo-nitzschia cells under a microscope, and the other half is chemically prepared to test for the presence of domoic acid, because again, not all species produce the toxin.

If the bloom gets big enough, and toxic enough, it poses a problem to Rhode Island’s shellfisheries, which would have to close immediately to avoid humans ingesting the toxin and developing Amnesic Shellfish Poisoning. The toxin can travel up the food web this way as the shellfish filter the water, eat the Pseudo-nitzschia,and accumulate their domoic acid, where it then builds up in humans who eat several shellfish consecutively. For this reason, we also monitor the mussel populations of the Bay for signs of domoic acid build up. Mussels filter water at a much greater speed than other shellfish like clams and oysters, and therefore are likely to be affected first. Mussels are also collected biweekly from three set locations covering the East and West sides of the Bay, dissected back at the lab, and analyzed for domoic acid. The three sample sites are rocky, but provide amazing views of Rhode Island, which makes the job fun and reminds me why I actually enjoy the scraped knees as a token of good field work.

This project is ongoing and closely coordinates with the Rhode Island Department of Environmental Management, which monitors shellfish and phytoplankton. Results will help Rhode Island better anticipate HABs and possible shellfishery closures to ensure shellfish safety and mitigate economic impacts. These efforts add to oceanographic research regarding environmental conditions that may spark domoic acid HABs, conditions that may be applied along other coasts wherever Pseudo-nitzschia is found.
[hr][divider][/hr]
Use of Robotics in Scientific Monitoring
Gibson Leavitt – Roger Williams University
SURFO advisor: Christopher Roman
In coastal towns, such as Narragansett, oyster farming is very important to thesociety and economy, and oyster farmers are increasingly more concerned about the quality of the water that the oysters are being grown in. Certain environmental factors can have massive implications for the benefitsand risks of growing oysters in a body of water. The oysters “breathe” dissolved oxygen (DO) and feed on particles in the water, commonly referred to as suspended particulate matter (SPM). Looking at temperature, DO, and SPM can indicate how efficiently the oysters can grow. These water quality parameters can also be related to diseases and harmful algal blooms that are dangerous to the oysters and the people who eat them.
We already monitor water quality on oyster farms, but the way we do it could potentially be unreliable. Buoys with sensors are placed in the water, constantly measuring the quality of the water beneath them. This means that datasets are either comprised of measurements in a few select places around a farm, or a series of measurements as the buoys flow downstream. To understand why this is a potential problem, imagine it’s raining wherever you are, which we will call point A. Now imagine that it’s also raining 30 miles away, at point B. Can you say for certain that it’s raining the entire distance in between both points? That’s the issue encountered by stationary data points. What if you decided to find out by biking the distance from A to B? Well, you would know if it was raining in between, but by the time you finished the bike ride, who knows how the weather has changed back at point A. This is a potential problem with relying on the current to carry sensors. But, what if instead you got in a car and drove to point B, then back to point A, and repeated this a few times? Then would you be able to say with some certainty that it is, or isn’t raining the entire distance between points A and B
Introducing: The Kayak
This latter method with the car is comparable to what we are trying to accomplish in water quality monitoring using a robotic kayak that acts as an autonomous surface vehicle (ASV).

The robot consists of the body of a kayak, but the seat holds 4 batteries to power the kayak and a computer that controls everything. Also included in the kayak is an electric motor to propel the kayak, 3 sensors attached to the side to monitor the water quality, and an antenna to communicate with another computer on shore. From shore, we are able to send missions to the kayak to tell itwhere to go and what to do. We can also monitor the kayak to make sure nothing goes wrong during a mission.
Unlike humans, robots never get bored or tired, so we can make the kayak repeat one path until the end of its battery life. An average mission for a kayak consists of running it in circles around an oyster farm for about 4 hours. This allows us to look for variation in water quality over both time and space, presenting researchers with more detailed data for research in the future.
In addition, this tricked out kayak can be rigged with a sensor to analyze water depth, also called bathymetry, and create maps of local salt ponds. Bathymetric maps are useful to scientists and fishermen alike, but we don’t possess accurate maps of every pond in the area. The kayak is able to follow lines precisely and collect depths at specific locations. From those readings, water depth can be charted. And voila! Bathymetric maps are made.
The Future
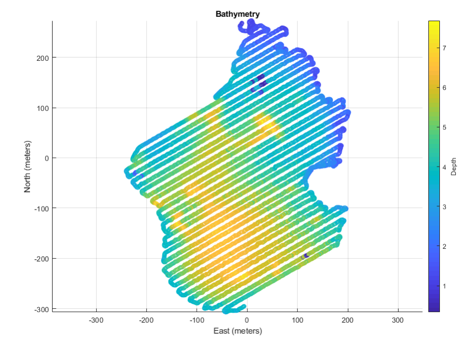
This specially designed kayak will allow for better data regarding water quality of local farms and potentially enhance the aquaculture industry of Rhode Island. However, more importantly, this kayak represents a future of using ASVs for water quality monitoring and a great example of the many potential benefits of unifying engineering and scientific monitoring.
I am a third year PhD student at the University of Rhode Island Graduate School of Oceanography in the Lohmann Lab. My current research interests include environmental chemistry, water quality, as well as coastal and seabird ecology. When not in the lab, I enjoy diving, surfing, and hanging out with my dog Gypsy.

