This is a post by undergraduate contributor Madeline Mamer. Madeline is a senior at the University of Washington in Seattle studying Earth and Space Sciences. She is passionate about cryosphere studies, specifically where ice meets the open ocean. In the future, she wants to use analog studies here on Earth to further our understanding of icy satellites like Europa and Enceladus. She drinks far too much coffee and on a Sunday afternoon, you can find her baking pumpkin cookies!
Article source: McConnochie, C.D., Cenedese, C., McElwaine, J.N., 2020. Surface Expression of a Wall Fountain: Application to Subglacial Discharge Plumes. J. Phys. Oceanogr. 50, 1245–1263. https://doi.org/10.1175/JPO-D-19-0213.1
The Background
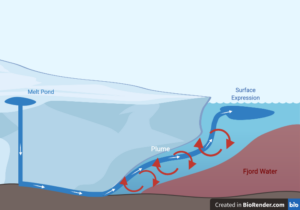
Think of the Bellagio fountains in Las Vegas, but put them in a fjord, surrounded by ice, and underwater. Huge fountains, combatting gravity, traveling upwards in the ocean, ultimately falling back down. This is just one of a few things that can occur when glaciers, or ice sheets, melt. Glaciers project streams, or plumes, of meltwater at their edge with immense force. A plume becomes a fountain when it has so much energy it travels upward to the water’s surface before it sinks.

But where does this water come from? This water is glacial melt that accumulates in pools on a glacier’s surface. The water can carve its own path or travel through preexisting caverns to reach the glacier’s edge. At the end of its journey, the meltwater is ejected as a plume into the surrounding environment.
In fjords where the ocean meets steep mountains, these plumes appear to be linked with glaciers that are experiencing faster than normal melting. The freshwater glacial plumes mix with the fjord’s warm, salty water. This mixing leads to an increased amount of warm, salty water hitting the glacier’s edge.
When these plumes become fountains, they create a shape at the water’s surface before sinking. Scientists want to better understand these shapes because this will allow them to infer what is happening at the glacier’s edge-such as where melting occurs and what that looks like. By having a better understanding of the glacial edge, scientists can gain a better idea of how glaciers are changing.
The Experiment
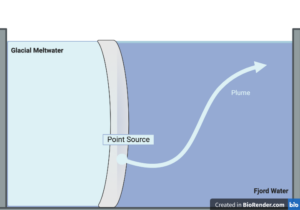
To piece together the relationship between the fountain’s shape and the edge of the glacier, Dr. Craig McConnochie and his team at the University of Canterbury set up a glass tank of water with a piece of acrylic plastic acting as the glacier. They drilled a hole into the center of the acrylic glacier to act as the source for the plume.
The tank was filled with a mix of saltwater and freshwater to recreate the normal conditions of a Greenland fjord. The scientists colored the dye that was supposed to be emitted from the point source in order to keep track of it as it traveled through the tank. A Nikon camera was positioned with LED lighting directly over the tank to record the whole experiment – basically an over the top YouTube cooking video.
They wanted to examine the fountain’s shape itself, as well as how quickly it spread out when it reached the surface. Looking at their recordings of the experiment, they discovered that the shapes ranged from semicircles to rounded triangles. Changing the angle of the acrylic allowed them to test the difference between a sloping glacier edge, and a vertical glacier edge. When the glacier edge was vertical, the surface expression spread out along the edge rather than spreading away from the glacier. They observed that the opposite occurred for sloping glaciers.
The Real-Life Scenario
To compare the results of their tank experiments with real-life observations, they gathered records from the Saqqarliup Fjord in Greenland. They discovered that the layers of water in the fjord are stable, like layers of a cake rather than the layers of a pudding. Because of this stability, their idea that the fountain comes to the surface, and then sinks, may not actually occur in the fjord.
They proposed a subduction event was happening, not a sinking one. During subduction, a heavier object slides under a lighter object. In the Saqqarliup Fjord, they were seeing the heavier surface expression sliding under the lighter water mass next to it. This apparent contradiction to their experiments occurred because in the lab set up their surface expression was like frosting on top of a cake, while in the Saqqarliup Fjord, the expression was like another cake layer. The difference in thicknesses between the surrounding water and the glacier’s plume changed the way the fountain interacted with the local environment.
This difference between the lab experiments and real-life observations goes to show how little we still know about what happens when a glacier meets open water. It demonstrates that current models and mathematical understanding which led the scientists to construct the lab setup still do not fully resolve what is happening in nature.
The Bigger Idea
Glaciers and ice sheets have a major responsibility here on Earth. Ice locks up water that would otherwise be in the ocean. Its bright surface reflects sunlight back into space, cooling the planet. The healthy glacial melt supplies ravines and lakes where precious ecosystems exist. These glacial fountains are just one component of a very complex system. Unlocking the secrets of plume shapes is part of a larger picture to understand how climate change is affecting glaciers and ice sheets. With the Greenland ice sheet reaching the melting point of no return and icebergs the size of New York City breaking off Antarctica, it is crucial we understand the complexities and magic of ice.
Kate received her Ph.D. in Aquatic Ecology from the University of Notre Dame and she holds a Masters in Environmental Science & Biology from SUNY Brockport. She currently teaches at a small college in Indiana and is starting out her neophyte research career in aquatic community monitoring. Outside of lab and fieldwork, she enjoys running and kickboxing.

