Baldassarre, L., Reitzel, A. M., & Fraune, S. (2023). Genotype–environment interactions determine microbiota plasticity in the sea anemone Nematostella vectensis. PLoS biology, 21(1), e3001726. https://doi.org/10.1371/journal.pbio.3001726
Most animals live together with microbial partners. Many symbiotic microbes—symbiosis means “living together”—have co-evolved with their hosts over time, and a healthy microbiome can provide nutritional benefits to the host or protect against disease and stress. Look no further than corals for a famous example of microbial symbiosis; the partnership between corals and photosynthetic algae forms the foundation of coral reef ecosystems. Like other animals, corals also host complex communities of bacteria, and scientists are just beginning to explore the factors that determine the composition of bacterial communities in corals and other marine animals.
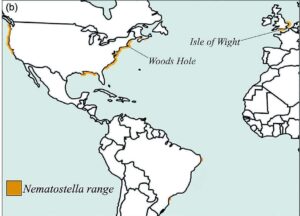
Two general possibilities exist for why animals host different microbial communities. A microbial community might be totally determined by the environment, so that different animals living in the same conditions would have the same microbiome. Alternatively, a microbial community might be specifically adapted to its host, so that the same animal would have the same microbiome in different environments. Scientists have found a great study system for marine microbiomes in Nematostella vectensis, a small sea anemone (a relative of corals). This worm-like critter lives in marshes and estuaries along the eastern coast of North America. Because of its wide geographic range (stretching from Florida in the south to Nova Scotia in the north), Nematostella is adapted to a wide range of environmental conditions, allowing researchers to study the impact of environmental factors and host genetics on the bacterial community.
A who’s who of anemone-associated bacteria
In this study, scientists sampled Nematostella from five different populations across the Atlantic coast, and across multiple seasons. They identified the different bacteria living in the anemones by sequencing their DNA. Geographic location was the most important factor in the composition of the bacterial community—microbiomes of animals from the same location were more similar to each other than to other populations. Temperature and salinity both contributed to microbiome differences, but temperature was more important.

The researchers also brought Nematostella from different populations into the laboratory in order to grow them in common conditions. After one month in the lab, the bacterial communities of the anemones became more similar to one another, and the overall diversity decreased (some bacteria were lost). However, anemones still maintained a microbial signature of their original geographic location! This means that differences between populations cannot be explained solely by the environment, but may instead reflect co-adaptation between hosts and symbionts.
“Is it hot in here, or is it just me?” – some bacteria, probably
To understand the effects of temperature in more detail, the scientists designed a follow-up experiment in which they took 12 different clonal lines from different locations—each genetically identical—and raised them at three different temperatures for three months. At the end of the experiment, temperature did indeed have a big effect on the bacterial community, but the different clones were still separate from one another. Interestingly, there was also an interaction between temperature and genotype, meaning that the effect of temperature on the microbiome depended on the clone.
So, after collecting all these data, does microbiome structure depend more on host identity, or is it determined directly by environmental influences such as temperature? Well, as usual in biology, it’s complicated! The microbial community depends on both the environment and the identity of its host. Some bacteria might have narrow environmental tolerances, or simply live “around” the anemone without evolving a close association. On the other hand, there is also a core microbial community that sticks with the host after moving to the lab, which likely co-evolved with the host.
The next step in this line of research is to study the function of different bacterial communities, because certain combinations of host and bacteria might differ in their tolerance of temperature or other environmental factors. These days, many biologists use the term “holobiont” to refer to the super-organism formed by a host and its microbiome. Humans are part of a holobiont—you, and all the trillions of bacterial cells in your body. This concept has huge implications for our understanding of ecology. For example, the ways that animals respond to climate change will depend not just on their own DNA, but on their interactions with microbial partners.
Cover image source: Trish Hartmann, via Wikimedia Commons

I am a PhD student at MIT and the Woods Hole Oceanographic Institution, where I study the evolution and physiology of marine invertebrates. I usually work with zooplankton and sea anemones, and I am especially interested in circadian rhythms of these animals. Outside work, I love to play trumpet, listen to music, and watch hockey.


