Source: Pohlman, J. W.; Greinert, J.; Ruppel, C.; Silyakova, A.; Vielstädte, L.; Casso, M.; Mienert, J.; Bünz, S., Enhanced CO2 uptake at a shallow Arctic Ocean seep field overwhelms the positive warming potential of emitted methane, Proceedings of the National Academy of Sciences 2017, 114 (21), 5355-5360. DOI: 10.1073/pnas.1618926114
It’s often hard to remember how methane, a gas of one carbon atom bonded to four hydrogen atoms, fits into the conversation on global warming. That is, at least until cows and sheep are mentioned. Ruminants, or mammals that acquire their energy by eating grass, release a substantial amount of methane into the atmosphere. Methane is a powerful greenhouse gas, trapping about 25 times as much heat compared to its more famous greenhouse gas cousin, carbon dioxide.
In addition to digestion by ruminants like cows and sheep, other processes that add methane to the air include production and transportation of natural gas and decomposition of landfill waste. But there are some less familiar natural sources of methane as well. One such source is melting ice; more specifically, frozen sediment called permafrost containing trapped methane, which bubbles out as the ice melts.
Thousands of years ago during natural climate warming events, rising sea levels flooded long expanses of permafrost along coastlines. The submerged permafrost remained frozen, keeping its methane stores locked in. But when the surrounding water heats up, permafrost begins to thaw and methane hydrate (a form of methane bound up as a solid) begins to break down into methane gas. As the Arctic warms (thanks to global warming), more of this subsea permafrost will melt, releasing methane into the atmosphere. Areas of methane release, referred to as methane seep fields, have been regarded as a net source of greenhouses gases. But what’s been ignored is the fact that in some cases this release of methane gas is accompanied by a substantial local drawdown of carbon dioxide – enough to even offset the effects of the methane.
Although big underwater air bubbles are easy to imagine when we try to picture gas going between seawater and the atmosphere, such large bubbles are not primarily responsible for exchange of gas between the two. Think more along the lines of the barely visible tiny bubbles in your favorite carbonated drink. In the same way that gases are present in measurable quantities in the air, gases can be dissolved into and have measurable concentrations in seawater. The fizziness in soda is simply carbon dioxide dissolved in the drink. Gas is constantly being exchanged between the air and the sea, and will eventually be exchanged until the air and the water have the same concentrations of that gas (also known as reaching equilibrium). If seawater has a gas in lower concentrations than is present in the atmosphere, the water will take up some of the gas from the air. Conversely, if the seawater has more of a certain gas than the air, it will release it to the atmosphere. This tendency towards equilibrium is what drives gas exchange between the ocean and atmosphere.
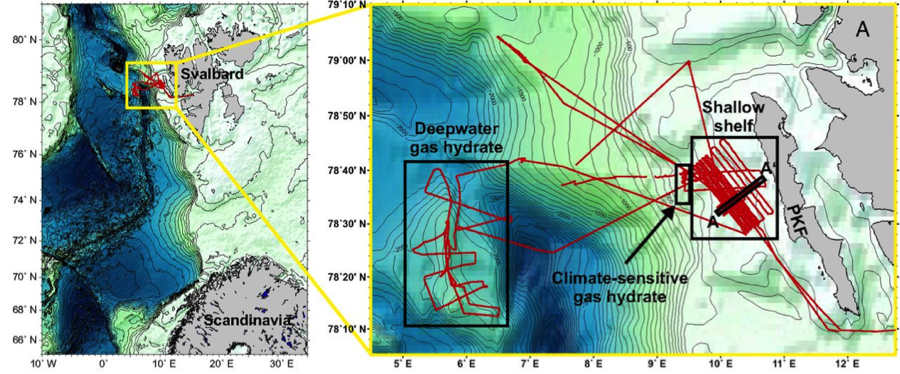
To settle whether methane seep fields were net sources or sinks for greenhouse gases, Pohlman and his collaborators examined the western Svalbard margin (Figure 1), off the Svalbard archipelago in Norway (home to the Svalbard Global Seed Vault, for the agriculturally inclined). This area presented an ideal test bed because it contains the full range of possible methane-emitting sites, including deep and shallow seep fields. They used measurements of methane and carbon dioxide (CO2) concentrations in the water along a 1,600 km-long transect through the region (Figure 1).
Despite there being many underwater sites that emit methane, it is rare for an appreciable amount of the gas to make it up to the atmosphere. Most methane released from the seafloor either dissolves or reacts with other chemicals during its ascent. From Pohlman and his colleagues’ measurements, it was evident that the shallow nearshore seep field was the only area where any of the methane was making it to the surface of the ocean (top left panel of Figure 2), as evidenced by the high surface water methane concentrations compared to the surrounding water. The measured emission of methane in this region ranged between 0.1 to 28.7 micromoles of methane per square meter each day (top right panel of Figure 2). To put these numbers in context, think back to the ruminants. For a 38.5 square mile region emitting methane at this rate, the daily methane release is equivalent to the daily methane output of about 320 sheep.
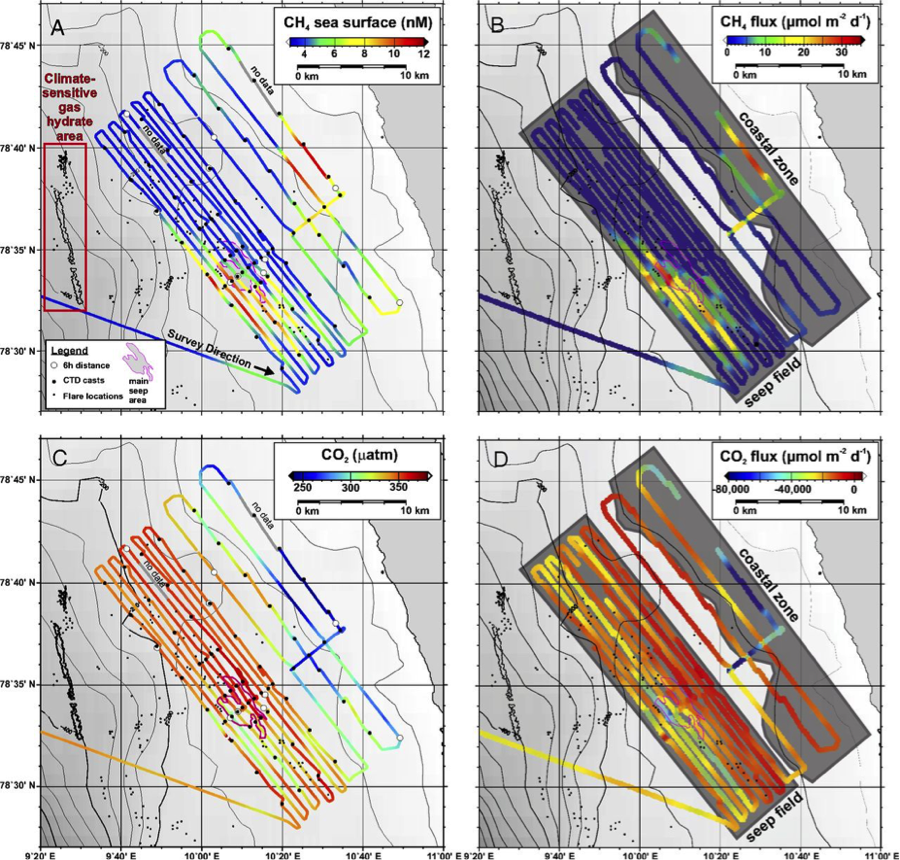
Not only was the water off the Svalbard archipelago relatively high in methane, it was also unusually low in CO2 compared to the water in the surrounding area (bottom left panel of Figure 2). These low seawater CO2 concentrations induce ocean absorption of CO2 at a rate of 33,300 7,900 micromoles of CO2 per square meter each day, which is more than 1,900 times greater than the rate of methane release (top right panel of Figure 2). Not all greenhouse gases are created equal, and as previously mentioned, methane traps about 25 times as much heat as carbon dioxide does. Even so, over a 25-year timescale, the cooling caused by the seawater’s CO2 drawdown in this region is 69 times greater than the warming caused by the release of methane.
So why is this high methane water low in carbon dioxide? The researchers explored two hypotheses: first, that methane bubbles forming at depths were rising to the surface, picking up carbon dioxide along the way, to eventually release both gases to the atmosphere. Their second guess was that something was stimulating phytoplankton photosynthesis, which uses up CO2 in the water. Pohlman and his colleagues played with scenarios in a seawater bubble model, but concluded that for the bubble hypothesis to be plausible, the rising bubbles would need to be at least 14 mm in diameter. The bubbles observed in the area were most frequently about 6 mm, not big enough to carry with it the necessary CO2 to explain the phenomenon.
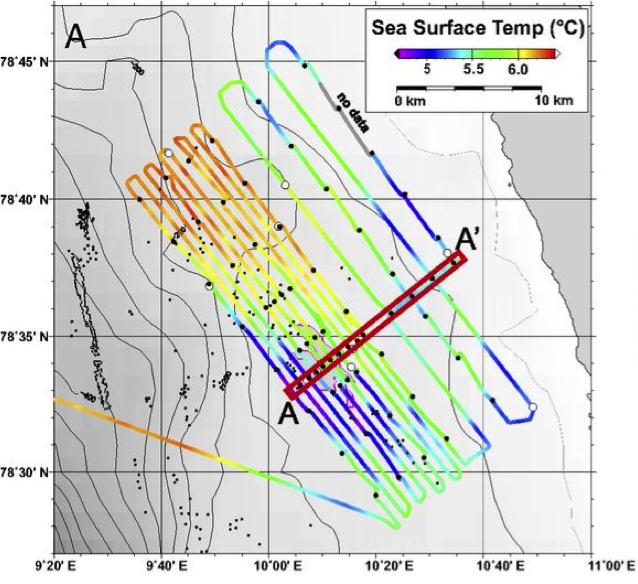
Their other hypothesis was that cold, nutrient-rich water was being brought to the surface, which would boost photosynthesis by phytoplankton. The water indeed had lower temperatures, higher nutrients, higher chlorophyll-fluorescence (a proxy for mass of photosynthesizing organisms), higher dissolved oxygen concentrations (a product of photosynthesis), and higher pH (also a result of photosynthesis when carbon dioxide is taken up). By comparing a map of sea surface temperature (Figure 3) to methane exchange (also referred to as methane flux) (top right panel of Figure 2), it becomes apparent that the low temperature waters coincide with the areas of highest methane flux. The same holds for the areas of carbon dioxide uptake. Finally, the CO2 uptake rates agreed with measurements of photosynthesis in a nearby region from a previous study.
This nutrient-rich water gets brought to the surface by wind pushing and pulling water around in a process called upwelling, which also likely helps the underwater methane make it up to the surface. Though bringing more methane to the surface is seemingly bad for the situation with global warming, CO2-deficient water being upwelled with methane may play an important role in offsetting the methane’s warming effects.
If low-CO2 water were not reaching the surface and the seawater did not absorb CO2 from the atmosphere, methane release would cause warming, further melting ice, causing sea levels to rise, further melting the permafrost, and finally releasing more methane. But instead in regions of upwelling such as off the Svalbard archipelago, enough CO2 is drawn down to quench this feedback, preventing a runaway situation in which warming was further exacerbated.
While the CO2 drawdown serves as a promising control on methane release from these underwater seep fields, questions still remain about the mechanisms causing methane release and CO2 drawdown. Although Pohlman and his colleagues’ work serves as a promising first study of net greenhouse gas exchange in methane seep fields, there remain numerous methane seep fields to be examined. Further research is needed to investigate these processes in other seep fields and whether this methane release will be offset by CO2 uptake in the future or in other locations.
Julia is a PhD student at Scripps Institution of Oceanography in La Jolla, California. Her focus is on biogeochemistry, which, as the name suggests, centers on the combined effects of biological, geological and chemical processes on the earth system. She is advised by Dr. Ralph Keeling and is modeling the global carbon cycle to better understand how much carbon dioxide ends up in the atmosphere. When not at her computer writing code, Julia can usually be found reading and/or thinking about food.


