Jayakumar A, Chang BC, Widner B, Bernhardt, Mulholland MR, Ward BB. 2017. Biological nitrogen fixation in the oxygen-minimum region of the eastern tropical North Pacific ocean. ISME Journal 11:2356-67. doi:10.1038/ismej.2017.97
Life in the ocean runs on nitrogen. Nitrogen is an essential building block for cells, needed for everything from replicating DNA to building proteins.
The good news is that nitrogen is everywhere is in the ocean. The bad news is that most of this nitrogen is in a form that cells cannot use – nitrogen gas. Nitrogen gas is highly stable, which from a cell’s point of view means that it takes a huge amount of energy to convert it into a form of nitrogen that they can use, like ammonium. In fact, this process is so specialized and energetically expensive that only a handful of bacteria can convert nitrogen gas to ammonium – a process known as nitrogen fixation. The result is that readily usable forms of nitrogen are highly prized, difficult to come by, and controlled by a small subset of microorganisms.
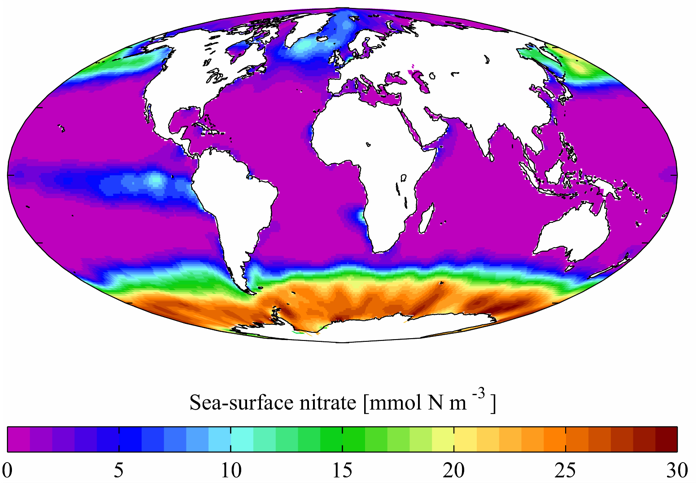
Nitrogen, in forms that cells can use like ammonium and nitrate, is scarce in most of the ocean. Nitrogen fixation is the primary source of readily available nitrogen in these regions. Photo credit: World Ocean Atlas.
Scientists are very interested in determining exactly how this delicate situation plays out since it determines just how much life the ocean can support. That, in turn, has far-reaching consequences for everything from the amount of greenhouse gas emissions that the ocean takes up, to the success of commercial fisheries, to the amount of oxygen on earth.
There’s just one major hurdle standing in the way of understanding how much usable nitrogen is in the ocean: all the measurements of how fast nitrogen fixing organisms actually fix nitrogen in different parts of the ocean may be wrong.
Until recently, a single procedure has been used to measure the rate of nitrogen fixation in a given patch of ocean. Scientists would sail out to wherever they wanted to sample, collect seawater into an airtight bottle, and then inject a tiny amount of a radioactive form of nitrogen gas into the water. In theory, that bubble of radioactive nitrogen gas would dissolve into the seawater and some of it would be converted to ammonium by nitrogen fixing bacteria and incorporated into DNA and proteins over a period of about 24 hours. The cells would then be collected at the end of the incubation so that their radiation dose could be measured, indicating how much of the added nitrogen gas they had converted into ammonium.
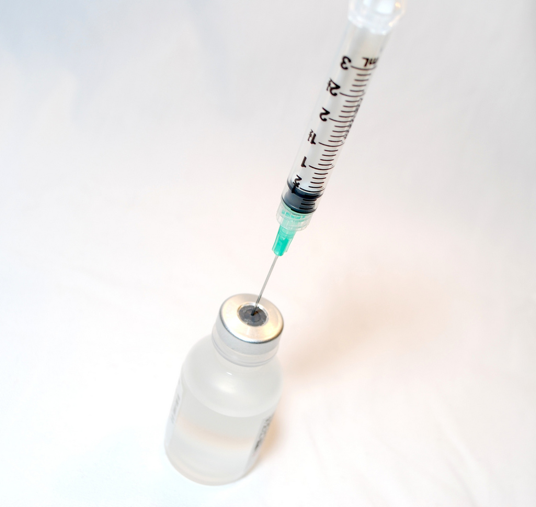
Researchers traditionally measure nitrogen fixation rates by injecting radioactive nitrogen gas into an airtight vial of seawater. Photo credit: National Institutes of Health
The problem, discovered only after decades of applying this procedure to measure nitrogen fixation rates all over the world, is that the added bubble of nitrogen gas doesn’t completely dissolve into the seawater. A portion of the added radioactive nitrogen gas remains in bubble form, inaccessible even to nitrogen fixing bacteria. The result is that the measured rates of nitrogen fixation are underestimated – by a factor of about 50%!
Now, realizing this mistake, a group of researchers has developed a modified procedure to more accurately measure rates of nitrogen fixation in the ocean. The change was simple: run the experiment as had always been done, but collect twice as much seawater and poison half of it. The poisoned bottle, treated with a chemical that would kill all nitrogen fixing bacteria, could be used to measure how much of the added radioactive nitrogen gas actually dissolves into the seawater during a 24-hour period. With that new denominator in hand, the researchers could calculate the nitrogen fixation rate in the bottle with active nitrogen fixing bacteria based on how much radioactive nitrogen gas they could actually access from the seawater.
The researchers put this method to the test in waters off the coast of southern California. Although readily available nitrogen is abundant in this region relative to much of the ocean, they were able to measure nitrogen fixation by bacteria. This indicates that despite the energy cost of fixing nitrogen, bacteria are fixing nitrogen even in regions where it may not be essential to for survival – a finding that opens up a new set of questions for scientists to explore.
While scientists now know that many of the nitrogen fixation rates that have been measured in the past are underestimated, the adjusted procedure developed here offers a solution going forward. As researchers learn more about how fast nitrogen fixation is occurring around the ocean, they can probe the delicate balance of oceanic nitrogen availability in more detail and better understand what controls the amount of life that various parts of the ocean can support
I’m a 5th year PhD student at Oregon State University researching the microbial ecology of marine sediments – why do we find microbes where they are in the seafloor, and what are they doing there? I spend my non-science time in the Cascade Mountains with my camera (@wanderingsolephotography) or racing triathlons.



I thought they used a stable isotope.