Collin P. Ward, Cassia J. Armstrong, Anna N. Walsh, Julia H. Jackson, Christopher M. Reddy. Sunlight Converts Polystyrene to Carbon Dioxide and Dissolved Organic Carbon. Environmental Science and Technology Letters, 2019. DOI: 10.1021/acs.estlett.9b00532

We humans have an addiction to plastic. Much of the plastic we rely on daily ends up in the ocean, where it lasts forever – or does it? A new study out of Woods Hole Oceanographic Institution suggests that some types of plastic may actually take far less time to degrade than previously thought, thanks to the magical powers of sunlight.
Plastic is Forever
Every day, each of us uses countless plastic items – from our first sip of coffee in a disposable cup, to the Tupperware containing our lunch, to even the clothes on our back. Plastic is one of the most common types of trash in the ocean, with an estimated 8 million metric tons – a weight equivalent to that of 90 aircraft carriers – entering the ocean each year. Some of this plastic floats around, littering our coastlines, damaging coral reefs, and entangling aquatic wildlife. Much of this plastic also breaks into tiny pieces called microplastics, which are ingested by marine animals, transferring toxic chemicals up the food chain.
In recent years, governments and companies have started to crack down on single plastic use – it’s why you now have to remember to bring your own bags to the grocery store, and why your Starbucks iced coffee now comes in a sippy cup, sans straw. These policies are based on the notion that plastic takes thousands of years to decompose, and therefore lasts forever as far as humans are concerned.
And this is true if we are only considering the length of time it takes microbes – tiny, living things such as bacteria – to “eat” plastic in the ocean and break it down. However, a team of scientists from Woods Hole had the bright idea that sunlight could break down plastic much faster than it takes microbes to finish their snack.

Light it Up
One of the most common forms of plastic is polystyrene, which is used in everything from yogurt containers to packing materials. It was the first plastic detected in the sunlit surface ocean back in the 1970s. The chemical structure of polystyrene contains a backbone of large, ringed molecules that microbes find hard to digest (picture eating a nice fillet of fish versus a whole bony fish), taking them thousands of years to break down completely. But what if other factors in the environment – say, sunlight – could break down polystyrene at a much faster rate?
To assess this hypothesis, the Woods Hole researchers tested five types of commercially-available polystyrene. Three were “pure” while the other two contained additives, which caused them to have slightly different physical and chemical properties. The scientists submerged the polystyrene in vials of seawater and subjected the samples to artificial sunlight using a special lamp called a solar simulator. As the simulated sunlight was 3 to 24 times stronger than natural sunlight (measured at the latitudes where the greatest plastic discharge to the ocean occurs), the scientists took 1 day of simulated sunlight to equal 3 to 24 days of natural sunlight.
After the samples’ tanning session, the scientists looked for signs of the breakdown of polystyrene into its basic building blocks. Chemical clues they sought included the production of carbon dioxide gas, and the formation of dissolved components in the seawater. Given that polystyrene is produced using petroleum carbon sources, they also kept an eye out for a petroleum signal in the dissolved components. During their detective work, the scientists used a number of sophisticated lab instruments, including a room-sized mass spectrometer.
Now You See It, Now You Don’t
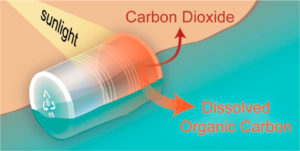
Lo and behold, the scientists found evidence of breakdown by sunlight in all of their samples. Apparently, the very thing that microbes find so hard to degrade, namely the ring-based backbone of polystyrene, is the perfect size and shape to catch solar rays. The energy provided by sunlight cleaves polystyrene’s carbon bonds apart, eventually causing the plastic to essentially disappear.
The scientists also found that the two samples containing additives actually broke down faster than their “pure” counterparts. As different additives seem to absorb different frequencies of sunlight, the exact formulation changes the rate at which the plastic breaks down. This adds yet another layer of complexity in determining the fate of plastics in the environment.
Take it With a Grain of (Sea) Salt

This lab experiment suggests that one type of plastic, polystyrene, might only take centuries to break down when exposed to sunlight, rather than millennia. Furthermore, it may only take decades for solar rays to partially degrade polystyrene. The broken down plastic is far easier for microbes to consume, meaning that a combined attack by sunlight and microbes might remove polystyrene even faster than estimated – although this hypothesis remains to be evaluated.
While this study is an illuminating first look into the important role sunlight may play in the breakdown of plastic in the ocean, it is important to interpret the results with a grain of salt. Keep in mind that the scientists investigated how only a handful of plastics – out of the innumerable types and formulations – behaved under the carefully controlled conditions of the laboratory.
For one, they used thin polystyrene films and small pellets in tiny vials of seawater, which may absorb light differently than “real” plastic in the environment. For another, the light exposure occurred over only a few days, and their estimate of a centuries timescale is exactly that: an extrapolation of their experimental results to calculate a theoretical lifespan – and the “real” environment rarely behaves as expected, as you probably experience on a regular basis with the weather forecast.

Regardless of its lifespan, plastic in the ocean will never be a cause for celebration. Even if plastic only lasts for decades, and not millennia, it still has devastating effects on the environment. The best way to reduce its harmful impacts is to curb the amount we discard in the first place. As a secondary precaution, these findings suggest that we can also work on smarter ways to make sure the plastic that does pollute our water spends as little time there as possible. And perhaps one day, we will no longer face an ocean drowning in plastic. ■
I am a Ph.D. candidate at Boston University where I am developing an underwater instrument to study the coastal ocean. I have a multi-disciplinary background in physics and oceanography (and some engineering), and my academic interests lie in using novel sensors and deployment platforms to study the ocean. Outside of my scholarly life, I enjoy keeping active through boxing and running and cycling around Boston.

