Undergraduates from all over the US have come to the University of Rhode Island Graduate School of Oceanography this summer to pursue research projects in oceanography as part of the Summer Undergraduate Research Fellowships in Oceanography (SURFO) program. Learn more about what they’ve been up to in this two-day series of short blog posts they’ve written for oceanbites!
“Finding a Stream in the Sea” by Keaton Brenneman
“What About Water Pollutants?” by Brittany Kerr
“Finding Phytoplankton: A 38 Year Journey” by Amanda van Buskirk
“Tremor in Alaska” by Blake Cross
“Guitar Strings of the Ocean” by Emma Thomas
“Finding Dissolved Oxygen with Foraminifera” by Felicia Rodier
Finding a Stream in the Sea
Keaton Brenneman — Rutgers University

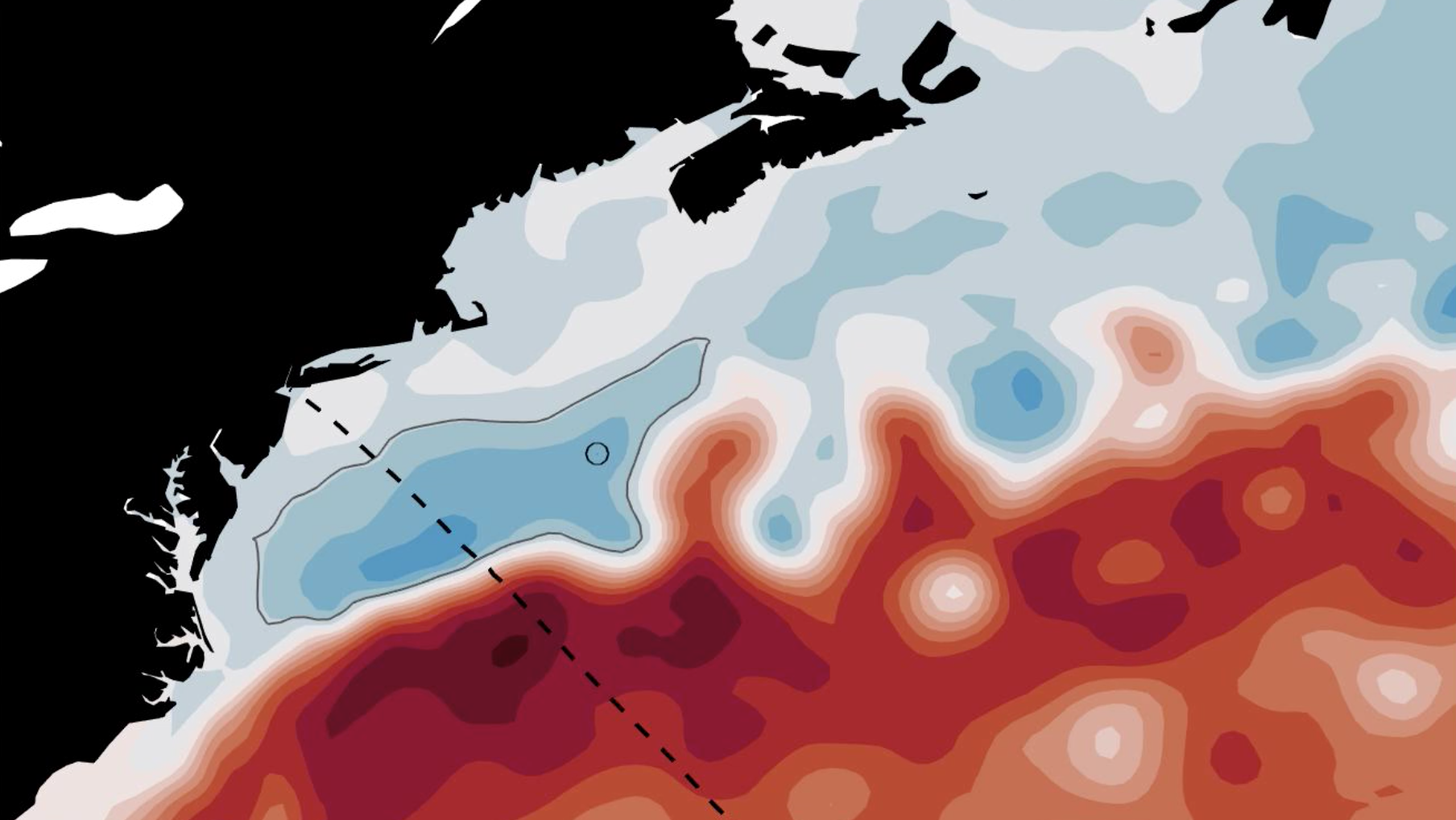
This summer for my SURFO project at URI GSO, I will compare real world Gulf Stream measurements with predictions of where the Gulf Stream is, how fast it is, and its structure that were made by NOAA’s Global Real-Time Ocean Forecast System (RTOFS). This comparison is invaluable to ocean modelers who need to determine possible sources of error in their models. Improving a computer model’s ability to resolve the Gulf Stream not only benefits the shipping industry and commercial fisheries, but can also lead to more accurate climate change predictions and European weather forecasts.
NOAA’s Marine Modeling and Analysis Branch is responsible for RTOFS, which creates daily predictions of the state of the ocean. These predictions forecast conditions up to 120 hours in the future and are updated daily for free use. Within NOAA, the model is used by the National Weather Service, the Tropical Prediction Center, Fisheries, Emergency Response Division, and many other branches. Being able to rely on these forecasts is very important for these organizations’ daily operation.

My project will focus on the Gulf Stream, which has been utilized by ships since the 1500s to speed up the trip from the Americas to Europe. It wasn’t until the late 1700s that Ben Franklin took the time to interview ship captains to determine the location of the Gulf Stream. Since that time, the shipping industry has utilized the Gulf Stream to more efficiently move goods across the Atlantic, and fishermen have looked at its location to determine where they’re likely to find certain fish.
Besides shipping and fisheries, the Gulf Stream plays a pivotal role in distributing heat from the equator to the poles, and affects weather in Europe. Great Britain’s mild, wet weather is due to the warm wet air above the Gulf Stream. If it weren’t for the Gulf Stream, Scandanavia would be a frozen wasteland! Any change in the amount of heat the Gulf Stream transports to Europe could have massive implications for Northern and Western Europe.
Recently, computer models of the ocean have been utilized by ship captains and commercial fishermen to plan trips. While these models can predict where the Gulf Stream will be in the future, their accuracy and precision must be evaluated by real-world observations. For the past 20 years a container ship, the CMV Oleander, has measured current velocities from 50 to 200 meters depth on its weekly trips from New Jersey to Bermuda. With this data, scientists are able to precisely determine where the Gulf Stream is and its properties.

What About Water Pollutants?
Brittany Kerr — Adrian College
There is a high value associated with organic, man-made chemicals. They can be found in many common everyday items in the form of flame retardants and pesticides, among others. On the other hand, there is a negative side to using pesticides and flame retardants as seen by the popular stories of these chemicals, such as DDT and PCB, leaking into the environment contaminating oceans, ice, and other bodies of water.
While we frequently hear about pollution and its damaging effects, we rarely hear about any efforts made to address this problem, prompting many questions. Are scientists even trying to solve the problem? Why are these chemicals still being manufactured if they are causing harm to the planet?
Scientists and manufactures are doing what they can with the resources they have. But the real issue is that scientists still haven’t fully grasped the problem in its entirety, and manufacturers are following rules set by the EPA while waiting on the scientists to discover potential problems.
Chemical manufacturers have a job to do: protect what people use and eat. Unfortunately that means creating chemicals that are beneficial for product stability and safety but that also may or may not pose a threat to the environment. Current EPA rules state that the chemicals are not considered harmful until they are proven guilty as such, which is problematic. In addition, scientists also need to understand the problems created by legacy pesticide and flame retardant chemicals, which are chemicals produced before the EPA stepped in to help control the production of these harmful substances, as well as the problems that may arise from the current-use pesticide and flame retardant chemicals. This is proving to take a long time.

For more than 20 years, researchers have looked into legacy pollutants to figure out how they got into oceans, polar ice, animals, freshwater, etc. and how long it took them to get there. They are constantly looking for more data and knowledge to shed light onto what the future may look like as a result of the use of these chemicals.
Scientists are also actively studying other types of pesticides and flame retardants to determine if they have contaminated our planet’s water. My research focuses on pesticides that are currently used in products today (referred to as CUPs) and flame retardants that are classified as organophosphate flame retardants (OPFRs) based on the dominant phosphate group within their structure.
To study these chemicals, we use a technique called passive sampling where a piece of polyethylene (PE) sampler, which looks like a piece of a plastic grocery bag, is deployed into the water of interest for a period of three to six weeks. The PE absorbs the organic pollutants present in the water, and we can then extract those chemicals to determine not only what they are but also their concentration within the water.
My work is to determine, in the lab, the ratio (which is called a partitioning coefficient) between the individual CUPs and OPFRs that were absorbed into a sampler versus what remains in their water samples. I do this by allowing a PE to absorb the CUPs and OPFRs from my own pollutant-injected water for a period of three weeks. After, I extract the pollutants from the PE and from the water and run the samples through a gas chromatograph and mass spectrometer instrument that separates the pollutants and allows me to determine their final concentrations in the water and PE. Using these concentrations, I calculate each pollutant’s coefficient, which can be applied to field research, allowing scientists to identify how much of each pollutant is in the body of water they’re studying.
Ultimately, this research aids in quantifying the severity of water contamination in regards to my CUPs and OPFRs. This will not only help scientists to determine if chemical manufacturers’ chemicals are causing problems but it will also help to quantify those problems. Once the problems are fully understood scientists can then begin looking for ways to combat them and find solutions.
Finding Phytoplankton: A 38 Year Journey
Amanda Van Buskirk — Monmouth University

When you take a look in a bay, there are a lot of things you might see. Rocks, sand, clams, fish, etc. But what about all of the microscopic organisms that you can’t see? Those are phytoplankton, and they are the base of the marine food chain. Though tiny in size, their impact on the environment is much larger than you could possibly imagine. In a single milliliter of water, there could be as many as 100,000 phytoplankton swimming around and providing food for other organisms. So for my research, I will be looking at a 38 year dataset from 1959-1997 that documents the abundance of phytoplankton in the Narragansett Bay and categorizes environmental factors that influence their growth.
This research is important for everyone, not just scientists, because phytoplankton is the basis of the marine food chain. They also produce a majority of the oxygen we breathe. Without it, life would not exist. It is important to monitor the blooming patterns and seasonal/annual changes to see how our ever-changing climate is affecting their growth. By researching how phytoplankton abundances are correlated to environmental factors, we are able to more clearly monitor issues such as toxic blooms. Harmful phytoplankton can poison fish and other seafood and in turn poison the human who eats them. So while they may be too small for our eyes to see, they can have a major impact on our health. The goal is to look at data from the past to try and predict what the future might hold.
Unlike many students who take on summer internships, I do not have a background in this particular field. I am a mathematics and statistics major at Monmouth University in New Jersey. While the common assumption is that math majors become teachers or work for insurance companies, there is so much more that can be explored. I am interested in blending my love of math with my passion for the environment, and this internship shows how this is possible. I am generating and analyzing graphs, finding patterns in data, and learning about phytoplankton species all at the same time.
The main focus of my research is the winter-spring phytoplankton bloom, which occurs in the bay between January and April every year. Once I document when the peaks occurred each year, I will look at which environmental factors correlate with peaks and drive differences in duration and starting time every year. These environmental factors include temperature, the amount of light in the bay, the amount of silica (an element required by phytoplankton species to grow) in the bay, and the amount of chlorophyll produced by the phytoplankton. I can look at how these factors are related and which ones are the main drivers of bloom change. After all of the information is gathered and analyzed, I will be able to determine how the bay has changed over time and how the phytoplankton are responding to those changes.
No matter what field you choose to study, you can benefit from having knowledge about the creatures of our Earth. So next time you dip your feet in the water, think about all of the tiny organisms swimming around and keeping life going all around the world.
Tremor in Alaska
Blake Cross — Colorado School of Mines

Think back through your early education. Chances are you remember at some point learning about earthquakes and plate boundaries. Terms such as “epicenter” or “subduction zone” may still hold some meaning to many people. When learning about how earthquakes occur we are given the image of two large rigid bodies trying to slide past each, getting caught on each other and stopping, and then violently losing their hold and rupturing past each other. This is indeed an accepted understanding of how plates may interact, but unfortunately, there are more factors at play in relation to fault activity. In the case of subduction zones (where one plate goes under another) things get more complicated as one heads deeper into the fault past the upper portion where earthquakes typically occur.
Recently it has come to be understood that in this specific portion of the fault there is somewhat of a transition zone between the locked zone in which earthquakes occur and the deep end of the fault where the plate smoothly slides and melts into the mantle (Fig. 1). It is in this transition zone that a phenomenon referred to as slow slip has been observed in several different parts of the world. These slow slip events are a slow and somewhat restricted movement of the plates past each other that often occur over the course of several weeks. Researchers believe that these slow slip events may be causing small seismic events, called tremors. My research is working to help prove or disprove this idea as much of the slow slip process is still not well understood.

In order to better understand this process it is necessary to understand the relationship between tremors and the slow slip events to determine if they are in fact correlated. The most difficult part of making this correlation is finding the tremors in seismic data as they are often not much larger than the typical background noise observed on most seismometers (the instruments used to detect seismic activity). Tremors can be recognized separately from earthquake as they behave differently, with a much more gradual start and much smaller peaks relative to the background noise (Fig. 2). Once tremors are found in data they can then be compared to the times when slow slip was observed (this is done by using GPS tracking of the movement) and it can be determined if the two are happening at the same time.
This research is interesting and important as it could be extremely beneficial for understanding the behavior of subduction zones and possibly providing a better idea of when an earthquake may occur. Slow slips have been associated with large earthquakes in several instances. These slow slip events may act as indicators for earthquakes to come. Researching the relationship between slow slip events and large earthquakes could be crucial for predicting and preparing for large earthquakes.
Guitar Strings of the Ocean
Emma Thomas — University of Massachusetts, Amherst
When water flows around long, cylinder-shaped objects, such as cables used to tether off shore wind turbines and oil rigs, the fluid being forced around the cylinder causes it to vibrate. These movements are referred to as Vortex-Induced Vibrations (VIV), which can become a problem when repeated vibrations weaken marine structures over time, potentially causing significant damage. Cables that tow fishing trawls, for instance, are subject to this effect due to being dragged through the ocean at considerable speeds.

It is important to understand the factors that control VIV so they may be avoided or properly prepared for in the future; to work towards this goal, I am constructing a three-dimensional visualization of the wake caused by such a cylinder in water. Previous research has revealed that VIV occur because of the phenomenon of vortex-shedding, or the process in which vortices (spinning columns of water) form in the wake of the cylinder. Vortices may be shed at different rates depending on the size of the cylinder and the speed of the water flow, and so the “shedding frequency” refers to the frequency at which vortices are created in a given situation.
In the video above, the gray circle represents a cross-section of an underwater cylinder.
Vortices can be seen forming in the cylinder’s wake in this computer simulation of VIV.)
In many ways, cylinders undergoing VIV may be compared to guitar strings (another type of long, cylindrical cable). When a guitar string is plucked, it tends to vibrate at a very specific frequency, producing a specific musical note. A frequency at which a string is built to vibrate is termed a “natural frequency” of the string; other objects, including cylinders subject to vortex-induced-vibrations, also have natural frequencies at which they tend to vibrate when disturbed. Hence, when the shedding frequency of vortices is close to the natural frequency of the cylinder, forces associated with the vortices are able to cause high-amplitude and potentially damaging vibrations. Extremely long cylinders such as cables used in the ocean have an almost infinite range of natural frequencies such that these vibrations are almost always present.

There are a number of factors that complicate the shedding of vortices and a cylinder’s movement. As the cylinder vibrates, for instance, the wake responds to each new motion, so that the forces responsible for vibrations do not follow a consistent pattern. The shape of the wake as it changes over time has been previously studied many times. One extremely useful method, called Digital Particle Image Velocimetry (DPIV) works by seeding water with small particles, illuminating the suspended particles with lasers, and tracking their movement as they are carried by the current. One problem with DPIV, however, is that it is only possible to record the motion of one two-dimensional slice of water at a time without using many expensive cameras simultaneously. This summer, my goal is to find a method for compiling many such slices, all recorded at different moments in time and at different locations along the cylinder, into a three-dimensional visualization of the cylinder’s wake.
Finding Dissolved Oxygen with Foraminifera
Felicia Rodier — Salem State University
Oxygen is essential to most forms of life and can restrict marine life when there is very little. There are some areas of the ocean that naturally have low levels of dissolved oxygen, but these places change over time. We may not have a written record of these changes, but we do have a record kept in an unlikely place, marine fossils. Foraminifera and trace elements found in their shells can be used to determine past levels of dissolved oxygen. As a participant in the SURFO program, I am trying to figure out if it is possible to use Iodine concentrations in foraminifera to determine dissolve oxygen.
Foraminifera are microscopic plankton that create a calcium carbonate shell, called a test, around their bodies while they are alive (Fig. 1). Once they die, those tests sink to the bottom of the ocean. The shells stay intact as they sink and become part of the sediment. While the organism is alive, trace elements are taken up by the tests. These elements stay in the structure after the organism sinks.
We pull up these sunken foraminifera in sediment cores and relatively date them. This gives us the approximate age of the foraminifera within a certain section of the core. I want to test the dissolved oxygen from 1000 years ago so I need to pick a segment of the core that is approximately that old.
Iodine and Dissolved Oxygen
To determine the dissolved oxygen in the time period these organisms lived in, scientists look at the elements taken into calcium carbonate shells. So far, researchers have examined elements such as manganese and nitrogen, but we think iodine may provide additional support to results produced from using traditional methods.
Why iodine? Iodine exists in two different oxidation states in the ocean called iodide and iodate. Iodate can be incorporated into calcium carbonate while iodide cannot (Fig. 2). The levels of iodide and iodate are relatively stable in the ocean but change depending on the level of dissolved oxygen. When the amount of dissolved oxygen in the water becomes low, iodate is reduced to iodide. This iodide cannot be taken up by the foraminifera, therefore the ratio of iodine to calcium in those foraminifera is low compared to the ratios found in water with higher dissolved oxygen levels.
So why bother understanding past levels of dissolved oxygen? As humans, we use oxygen to stay alive, as do most other organisms. Dissolved oxygen is important to aquatic organisms, and when it is not available, the ecosystem changes drastically. Looking at past dissolved oxygen levels can identify areas that consistently have low levels and help create predictions on future changes. Testing if dissolved oxygen can be determined with iodine could produce another method to support data that has already been collected by traditional methods.
I am the founder of oceanbites, and a postdoctoral fellow in the Higgins Lab at Colorado School of Mines, where I study poly- and perfluorinated chemicals. I got my Ph.D. in the Lohmann Lab at the University of Rhode Island Graduate School of Oceanography, where my research focused on how toxic chemicals like flame retardants end up in our lakes and oceans. Before graduate school, I earned a B.Sc. in chemistry from MIT and spent two years in environmental consulting. When I’m not doing chemistry in the lab, I’m doing chemistry at home (brewing beer).