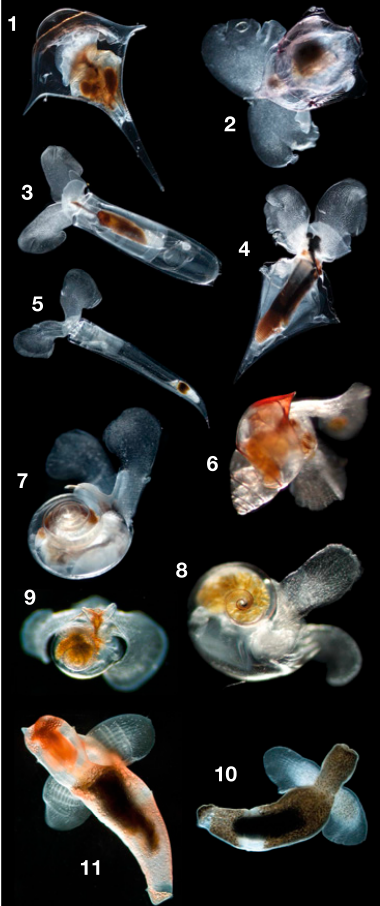
Many marine animals have a two-stage life cycle, with a free-swimming larval stage and a separate adult stage (examples include clams, starfish, and sea anemones). Although larvae are small and have simple body plans, they face the difficult task of navigating the vast ocean–and like the more complex adult animals, have sensory organs in order to do this. Many animal larvae have something called an “apical organ”, basically a clump of neurons and sensory structures at the front of the larva. These organs help the animal sense its environment and control its swimming behavior.
Since apical organs are found in the larvae of many distantly related animals, some scientists think they may have been present in a distant common ancestor. If that’s true, we would say these organs are “homologous,” or derived from a common ancestor. For example, the wings of a bat are homologous with human arms, but bat wings are not homologous with insect wings (because they evolved independently). Alternatively, larvae with this type of sensory structure may have evolved more than once, and our distant ancestors may not have had this structure.
Are the apical organs of animal larvae homologous, or did they evolve more than once? The answer may lie in an ancient group of animals called cnidarians.
Learning about larvae

Cnidarians include jellyfish, corals, and sea anemones. Cnidarians are very distantly related to most other animals (they split from our common ancestor more than 700 million years ago!), and unlike most animals, cnidarians don’t have brains—instead, they have a sort of “nerve net” that is not centralized. However, many cnidarians still have larvae with apical organs. Are they homologous to those of other animals?
Scientists can try to understand how organs evolve by measuring “gene expression.” This is a technique wherein scientists count the RNA molecules in a certain body part; genes with more RNA molecules have high expression and are probably important in that organ. For example, our eyes have high expression of genes needed for vision, and our stomach expresses digestive enzymes. If the apical organs of different larvae are the same, they should express the same genes even in different species.
Jellyfish in a league of their own
In this study, the researchers found genes expressed in the apical organs of 4 different species: two corals, one sea anemone, and one jellyfish. The coral and sea anemone larvae expressed similar genes in their apical organs, indicating that these structures are homologous. They also shared many genes with the larvae of other distantly-related animals such as sea urchins, again suggesting homology. However, the larval organs of jellyfish shared very few genes with the sea anemone and corals.
A window into the past
So, are the apical organs of animal larvae homologous are not? Larval gene expression is very similar between the sea anemone, corals, and other animals, suggesting that all these larval organs may be homologous. On the other hand, the jellyfish larva had few genes in common with other species. These data suggest that a distant animal ancestor may have had larvae with an apical organ that gave rise to the larvae in sea anemones and things like starfish and oysters—but that jellyfish, specifically, may have lost this structure. However, we will have to study more species in order to be confident in this answer.
Studies like this one allow us to see into the deep past. Comparing animals that live today lets us make informed guesses about what animals might have looked like hundreds of millions of years ago. This is especially useful for things like larvae, which are small, soft, and don’t leave many fossils. Although we still have a lot more to learn, we now have some evidence that an ancient animal had a larva with an apical organ, much like many animals living today. And jellyfish? Well, they may have gone down their own path.
Cover image source: Luc Viatour via Wikimedia Commons

I am a PhD student at MIT and the Woods Hole Oceanographic Institution, where I study the evolution and physiology of marine invertebrates. I usually work with zooplankton and sea anemones, and I am especially interested in circadian rhythms of these animals. Outside work, I love to play trumpet, listen to music, and watch hockey.