Reviewing: Peijnenburg, K. et al. The origin and diversification of pteropods predate past perturbations in the Earth’s carbon cycle. 1–9 (2020). DOI: https://doi.org/10.1073/pnas.1920918117
It’s a bird… It’s a plane… Its a pteropod!
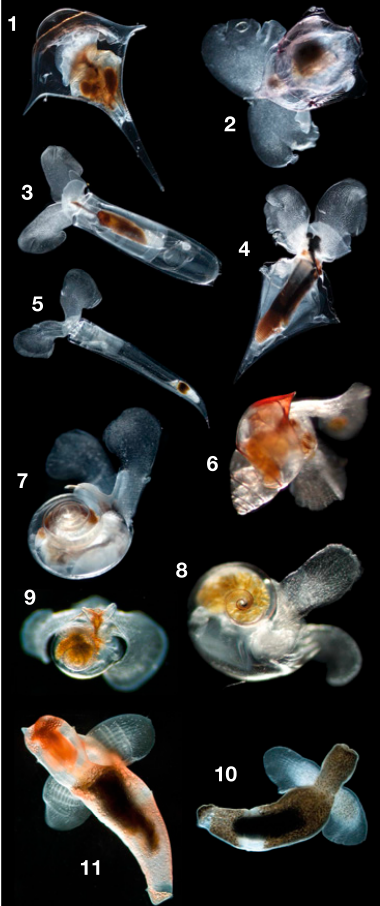
Have you ever heard of a flying snail? Meet the pteropod (with a silent “p”), or “wing footed” sea snail. These snails are considered pelagic meaning they spend their entire life inhabiting the open water rather of creeping along the seafloor. Pteropods and other zooplankton, such as krill, copepods and larvae, serve as a major food source for larger open-water fauna, like fish and whales. Over evolutionary timescales, these snails have thinned, or entirely lost, their shells to become lighter and morphed their foot into two wings for graceful swimming. Those without shells are called gynosomes, or sea angels, and the shelled varieties are referred to as sea butterflies.
An introduction to pteropods, or “wing-footed” sea snails. Courtesy of Monterey Bay Aquarium.
With climate change, excess CO2 dissolves into the ocean resulting in the water becoming more acidic. Molecules in the ocean, like calcium carbonate (which the sea butterflies use to build their shells), naturally buffer, or stabilize, the ocean’s pH (a measure of acidity) by chemically reacting with the CO2 molecules. However, human-derived levels of CO2 are so high that there is a significantly lower availability of these buffering molecules for animals who rely on them.
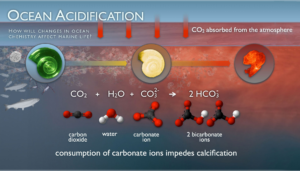
Short-term lab-based experiments where shelled pteropods are exposed to future concentrations of CO2 show a decreased ability to build shells resulting in thinner shells that dissolve quickly and even cause mortality. These stark results have caused concern amongst many researchers that pteropods, like many other animals dependent on calcium carbonate ions, may be at a risk of extinction due to ocean acidification.
Unlike the gradual process of ocean acidification in nature, lab-based experiments often do not provide adequate adjustment periods for animals due to time constraints. Hence, results from lab-experiments may instead measure animal’s response to a sudden change rather than how they will adapt to ocean acidification over time. Nonetheless, the best long-term experiment on survival is evolution for which we lack information on pteropods. Questions such as “when pteropods originated?”, and “how they have evolved over time” can provide better long-term evidence of their survival and insights into how they may fare in the next century against acidifying oceans. These are the questions that Dr. Peijnenburg and colleagues sought to answer.
Reconstructing the past
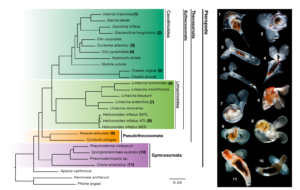
Pteropods were first identified in the fossil record in the Campanian (~72 million years ago). However, they don’t appear abundantly until the Paleogene (~56 million years ago). The fossil record for pteropods is particularly tricky to reconstruct. Gynosomes, those without shells, don’t preserve well, if at all, and those with shells often end up dissolved or damaged after sinking to the deep ocean. To supplement the fossil record, scientists reconstruct evolutionary pathways by mapping species onto a tree (specifically called a phylogenetic tree) where the root represents a common ancestor and the branches represent related species. For their research, Dr. Peijnenburg and her team collected pteropods, both shelled and unshelled, from the Atlantic Ocean and extracted the DNA of all the species. They used software to build a phylogenetic tree from the pteropod DNA and calibrated it to the fossil record time-stamping the branches to allow for interpretations relative to geologic events.
What did they find?
Dr. Peijnenburg and fellow colleagues found that the two major pteropod groups, the sea butterflies and sea angels, branched from seafloor-dwelling snails and slugs in the early Cretaceous period, approximately 139.1 million years ago. This date far precedes the earliest recorded fossils. If pteropods diverged this early it means they have survived multiple major global perturbations and associated extinction events. Notably, pteropods survived the Cretaceous–Paleogene mass extinction (or K–Pg boundary). You know, the one where a giant asteroid likely hit the Yucatán peninsula and wiped out 75% of species (including all non-flying dinosaurs) from the direct meteor impact or its secondary affects (like acid rain, ocean acidification, or food web collapses). That’s the one. What about the Paleocene–Eocene Thermal Maximum (PETM), a major shift in Earth’s climate about 56 million years ago considered analogous to modern-day ocean acidification? Pteropods survived that global climate change event too!
Does this mean pteropods are “safe”?
If pteropods showed resilience to major past climatic perturbation, they could survive current climate change, right? Not necessarily. Because the fossil record for pteropods is incomplete due to their lack of shell or fragile shell, we will never know how many species went extinct from these events and are simply absent from the fossil record. Additionally, the evidence that pteropods have survived major extinction events does not guarantee they will withstand the current extinction event caused by humans, the Holocene extinction event. This uncertainty rests on the fact that our current CO2 production rate far exceeds any record from the last 66 million years by at least 10 times. The authors suggest that current global changes are unlike anything any animal has experienced before in speed or intensity. Hence, we can’t expect pteropods to be resilient to anthropogenic climate change.
I am a recent PhD grad in Biological Oceanography at the University of Hawaiʻi at Mānoa. I study the community ecology of abyssal animals across space, time, and habitat, and I ask questions often with regard to human impact, specifically deep-sea mining.