Article: Unexpected source of Fukushima-derived radiocesium to the coastal ocean of Japan. 2017. Sanial, Buessler, Charette, and Nagao. PNAS. DOI:10.1073/pnas.1708659114
There are almost 450 nuclear power plants in the world. These plants use nuclear fission (splitting of atoms) to heat water, which produces steam. This steam moves giant turbines and produces energy. Nuclear power plants produce large amounts of energy without using fossil fuels, which produce greenhouses gases that contribute to global warming. After being built, they are also relatively cheap to maintain and run. However, they are also potentially dangerous when improperly managed, and major accidents can lead to nuclear meltdowns and contaminate large areas with radioactive materials. These materials can cause cancers, genetic mutations, and birth defects in living things, including humans.
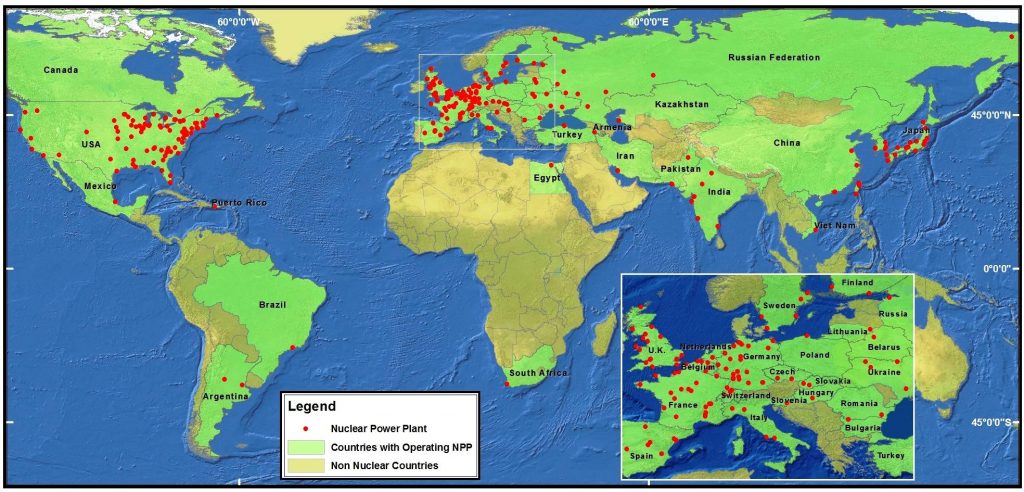

About half of these nuclear power plants are in coastal areas, making nuclear safety a concern for marine policy makers and conservationists. In 2011, a huge earthquake (magnitude 9) rocked the coast of Northeastern Japan. This earthquake caused a tsunami up to 20 feet high to crash against the Japanese coast. The Fukushima Daiichi Nuclear Power Plant was hit by this tsunami, resulting in nuclear meltdowns and release of radioactive materials.
One of the radioactive materials that was released was cesium 137, or 137Cs. 137Cs is an isotope produced through fission, i.e. breaking, of radioactive Uranium, in places like nuclear reactors. After the Fukushima meltdown, rivers and air were monitored for 137Cs. The highest concentrations of this atom were released immediately after the accident, and low levels of cesium are still being released from the accident today. Most of the escaped cesium made it into the ocean, either from rivers, from transport in the atmosphere, or from groundwater running directly from the accident site. Or at least that is what we thought, as these avenues were the only ones measured for radioactive cesium after the event.

A group of scientists in Massachusetts and Japan found a new source of 137Cs to the environment after the Fukushima accident. From 2013 – 2016, these scientists visited 8 different beaches within 100 km of the accident site. They took sand and water samples, which they measured for radioactivity using equipment that detections gamma radiation. Gamma radiation is a type of radiation in which photons are produced, and this type of radiation can pass through human bodies, causing all sorts of genetic problems. Scientists here were measuring the gamma radiation because 137Cs emits this type of radiation, so you can detect 137Cs by its distinctive gamma radiation patterns.
The scientists were also curious about how 137Cs interacts with the sand on these beaches. Sand adsorbs particles, like radioactive cesium, and can hold onto it. Eventually these particles will desorb from the sand, and be released back into the environment, like into seawater. The scientists did experiments where they tested how much 137Cs would adsorb onto the sand particles and stick there by flowing water with extra 137Cs through sand, and measuring how much 137Cs filtered through. They also used clean seawater to test how much 137Cs would ‘unstick’ from the radioactive sand samples by percolating the water through the sand for 4 hours.
The scientists found that at the bottom of their sand samples, up to 2 m deep into the beach, the 137Cs concentrations were incredibly high. They measured up to 480,000 Bq per square meter (Bq is a measure of radioactivity) in their deepest samples. The soil in the restricted zone of the Fukushima site, up to 100 km away, measured ~100,000 Bq per square meter. This means even tens of kilometers away, the radiation can actually be higher than near the disaster. They also measured 137Cs in rivers and seawater, which were thousands of times lower concentrations, meaning that the 137Cs must be coming from the sand, and not the surrounding water.
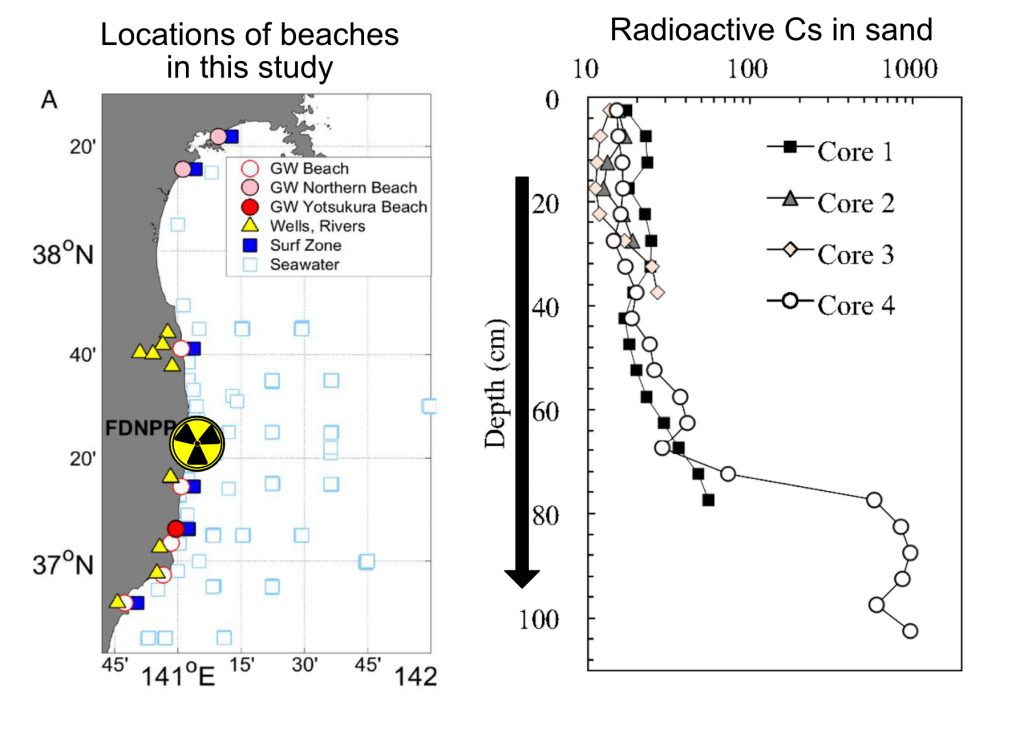
The sand adsorption and desorption experiments supported this hypothesis. These experiments showed that 99% 137Cs stuck to the sand during flowing through it. These experiments also showed that these radioactive atoms would come loose from the sand into water. Calculations and modeling performed by the researchers concluded that from 2013-2016, the amount of 137Cs released from sands into the ocean is similar to the amount of 137Cs released from underground the Fukushima disaster site itself during this time period.
How did this much 137Cs get into these sands? The scientists proposed that immediately after the Fukushima accident, ocean waves carried 137Cs along the Japanese coast. This radioactive mixed into beach sands, where the 137Cs adsorbed onto sand particles. In the years after the event, the 137Cs washes off the sand and into the ocean water, bringing more 137Cs into the ocean over a long time period (Figure 5).
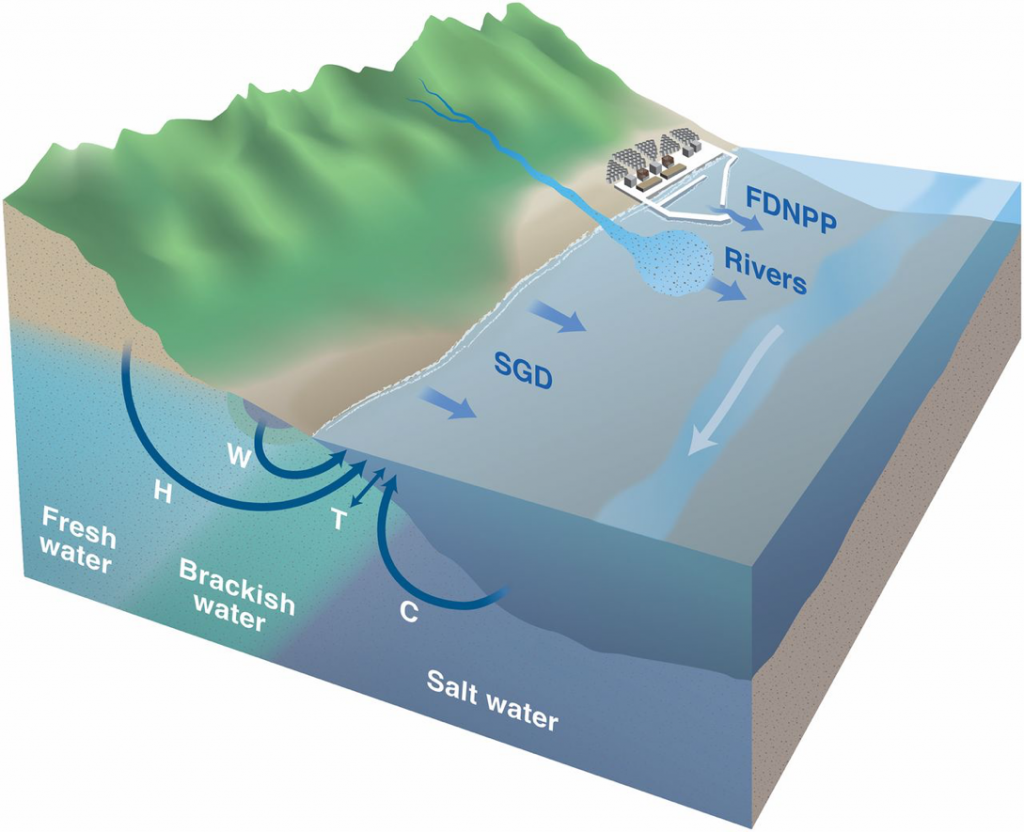
What should we take away from this study? The authors point out that this major reservoir of radioactive materials in the sandy beaches need to be considered in future nuclear management strategies. With proper planning for these disasters, the environment can be protected and harm to environments, including the ocean, can be mitigated.
I am a PhD student studying sediment geomicrobiology at the University of Southern California. My primary research interests lie deep under the sea studying how microorganisms survive in dark environments and how they interact with chemical cycles in sediment and on earth. When I surface from my studies, I enjoy backpacking, trying to mimic my ridiculous dog, and applying my laboratory techniques in the kitchen.


Have you seen what you have done to my face?