Each summer, the University of Rhode Island Graduate School of Oceanography (GSO) hosts undergraduate students from all over the country to participate in oceanographic research. These Summer Undergraduate Research Fellows (SURFOs) have not only been working with GSO scientists, but they also have spent part of their time learning how to communicate this science to the public. Although their research experience was virtual this summer, they still did a fantastic job. Read on to find out what they have been up to, and why they everyone should be as excited as they are about their work.
Tobias Kochenderfer is a Chemistry student entering his senior year at the University of Arizona in Tucson, Arizona. He is interested in analytical chemistry, chemical instrumentation, climate chemistry, geochemistry, and ocean engineering. His advisor this summer was Dr. Rainer Lohmann.
— — — — — — —
According to the Environmental Working Group (EWG), even months before entering the world, American babies are exposed to more than 200 chemicals in their blood, a majority of which are cancer-causing. Test a random person you see on the street’s blood and you will be surprised (and likely disgusted) with what you find! One of the most notorious classes of such pollutants are Per- and Polyfluoroalkyl substances, or PFAS for short.

These substances are common manufacturing aids in many “non-stick” products and are extremely pervasive pollutants due to their tricky chemical nature. They are found in the soil, water, and air and have notable health effects as outlined by Nathaniel Rich’s The Lawyer Who Became DuPont’s Worst Nightmare (https://www.nytimes.com/2016/01/10/magazine/the-lawyer-who-became-duponts-worst-nightmare.html). Tackling the PFAS problem is no small feat, considering there are over 4700 available PFASs—some of which are banned from manufacturing use, some that should be banned from manufacturing use, and some that are… passable. So, from a legislator’s perspective, how should they create a policy and identify the most delinquent of the thousands of PFAS?
In the early 2000s, 3M, a major PFAS manufacturer and others phased out several simple PFASs. More were eventually banned by the US EPA in 20151. That brings us present day, where several alternative PFAS structures are in manufacturing use; several of which are don’t break down in the environment and have many toxic effects1. These disappointing alternatives have ultimately led to the tactic of grouping when addressing the PFAS problem.

Why do we group?
Though easy and reasonable to claim that all PFASs are bad, it is worthwhile to discuss the benefits of PFAS-aided manufacturing. Water-resistant gear and firefighting-foam industries are two huge PFAS users, though are undoubtedly life saving. Somewhere in between the PFAS-saturated world we live in now and the environmentalist PFAS-free ideal lies the answer, and why grouping is needed.
Grouping PFASs allows for a more approachable means to address the sheer number of pervasive pollutants. Beyond this, grouping can minimize animal testing, inform consumers of their products, and address contaminated sites1. But how is grouping is conducted. Let us start with approaches based on properties based on the chemical nature of PFASs.
First there’s the P-sufficient approach. The P stands for persistent. Any PFAS that will eventually transform into an stable end product that remains in the environment for a long time is labeled as persistent1. This is a relatively straightforward grouping method but there is no legal precedent for this.
There is also a Bioaccumulation approach, which is based on the bioaccumulation potential of a given PFAS. To explain bioaccumulation, think of a tiny fish. They ingest a pollutant such as PFAS. The next predator up the food chain eats this fish and many other like it. The concentration of the pollutant in this predator is then far greater than the concentration in the original fish. This process continues up the food chain until PFAS is extremely concentrated in a large fish such as tuna. If a human eats an excess of large fish, they will likely grow sick. Bioaccumulation potential is hard to determine for PFAS due to its chemical structure and tricky behavior. A similar method that is quite powerful in scope when paired with the bioaccumulation approach is the Contaminated water approach. This method is mostly concerned with the potential of various PFAS to contaminate water resources.
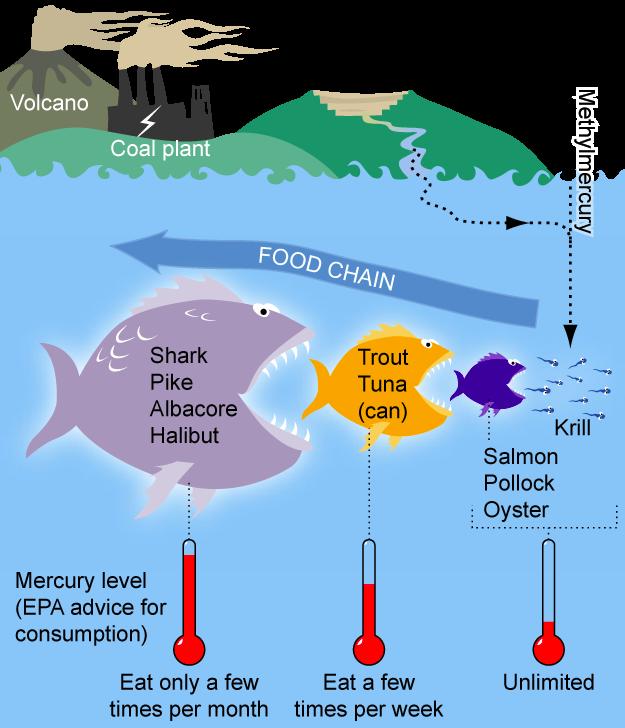
When substances dissolve more readily in water, it has a higher potential for moving through water sources to drinking water. When addressing plastics, there’s the PLC approach. There exists several types of plastics which are commonly used in various industries that involve PFAS in their manufacture. These can be classified as Polymers of Low Concern if they have are chemically inactive and contain no trace of PFAS in the plastic itself1.
There are also methods of grouping that categorize based on harm and risk involved with PFASs. First there is the Arrowhead approach. This method looks at the fate of any given PFAS. If it degrades into any already banned or dangerous PFAS it is considered as equivalent to that PFAS. This approach is the dominant approach to group PFAS for risk assessment and management1.
The Total organofluorine approach looks at manufactured goods rather than the substances themselves. For example, if I had a rain coat which I could feasibly dissolve, I could test the total fluorine content of the solution using various instruments which would be indicative of the fluorine content of the coat. Let’s hope its not above the regulatory levels!
The Simple additive toxicity approach correlates the toxic effects of the most notorious of a group of PFAS structures to the entire group. For example all PFASs that look chemically similar will be addressed as if they have the same toxic effects as PFOA1, a famous PFAS. A limit will then be set for the total concentration permissible in a given environment.
Similarly, the Relative potency factor approach groups similar structures as similarly concerning but weights their risk with their abundance1. The final method of grouping based on risk groups only PFAS with similar adverse effects, mechanisms of action. This is the most resource-intensive method of grouping. This risks the potential that few substances will actually be grouped into clusters of significant number1. This method will only have a marginal improvement on risk assessment1.
The applicability of each method largely depends on context. So let us consider our ambiguous legislator as we did in the beginning. They’re from a small riverside town whose residents have had a notable increase in kidney issues. There is no manufacturer which uses PFASs nearby, but many PFAS products have been discarded and found in the local river. The legislator may then follow the PLC grouping approach to address these concerns. For this context, and several others grouping provides a means to streamline the process of improving environmental and public health by restricting PFAS in manufacturing.
This last summer I have been working as a virtual intern in the SURFO program at University of Rhode Island’s Graduate School of Oceanography under the mentorship of Dr. Rainer Lohmann, Dr. Jitka Becanova, and Matthew Dunn. I am particularly interested in PFASs’ tendencies toward various environments. For example, water to air, water to plastic, etc. A more comprehensive understanding of the behavior of PFASs can both advise policy and progress the marginal documentation of arguably one of the United State’s most dire pollutants.
No doubt is the PFAS problem a nearly insurmountable issue. From a scientific perspective, PFASs have properties that make them difficult to study. From a public health perspective, PFASs are extremely pervasive and have numerous health hazards. And from a political perspective, legislation is and will be an uphill battle considering the utility of PFAS in manufacturing and the impressive technology that they help produce. Such impossible questions lend themselves to the practice of science. Where painstakingly procedural experiments are performed and reproduced to slowly build a solid foundation of understanding of which will be used to reach such unattainable goals.
1Cousins, I. T., DeWitt, J. C., Glüge, J., Goldenman, G., Herzke, D., Lohmann, R., Miller, M., Ng, C. A., Scheringer, M., Vierke, L., & Wang, Z. (2020). Strategies for grouping per- and polyfluoroalkyl substances (PFAS) to protect human and environmental health. Environmental Science: Processes & Impacts, 22(7), 1444–1460. https://doi.org/10.1039/d0em00147c
I’m a PhD student in the Rynearson Lab at the University of Rhode Island (URI) Graduate School of Oceanography (GSO). My research interests are focused on human impacts on the oceanic ecosystem, particularly effects on the primary producers (phytoplankton) at the base of the food web. Currently, I work with cultures from regions of the ocean that are nutrient limited and will conduct experiments to investigate how these phytoplankton survive.
