Source: Zhang, J., Liu, Z., Brady, E. C., Oppo, D. W., Clark, P. U., Jahn, A., Marcott, S. A., Lindsay, K (2017), Asynchronous warming and δ18O evolution of deep Atlantic water masses during the last deglaciation, PNAS, 114 (42) 11075-11080. DOI: 10.1073/pnas.1704512114
A fun but little-known fact is that we’re currently in an ice age. Having a hard time believing it? Pull up any picture of our earth and take a look at the poles. An ice age is defined by the presence of ice sheets in both the northern and southern hemispheres. The reason for the disconnect between our current climate and most people’s impression of ice ages is that we’re living in an interglacial period – a time when ice sheets have retreated (relative to glacial periods, when they’re at their furthest extent) (Figure 1).
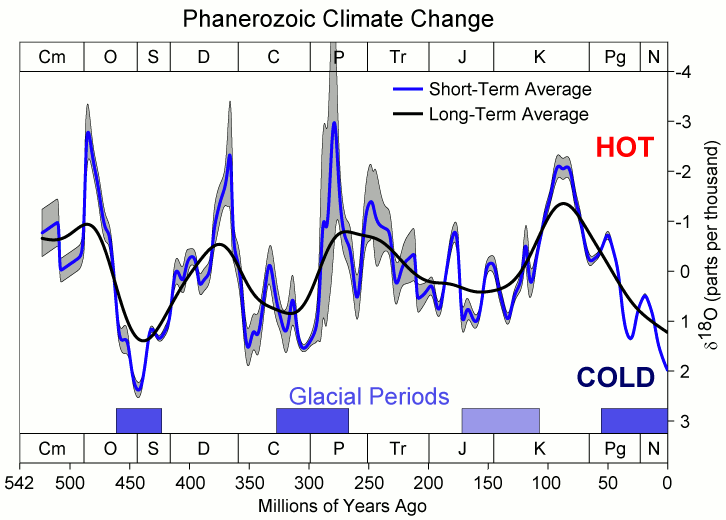
The wonderment of living in an ice age is all the greater when considering the forces at work that moved our planet into its current state. In the absence of humans emitting fossil fuels, the state of the global climate is pretty resistant to change. But every 100,000 years or so (Figure 2), some minor (and still unexplained) kick can send our planet into a period of transformation until we reach a new and profoundly different state, which, in our case, is the current interglacial period.
All sorts of things changed during this transition from the Last Glacial Maximum (furthest ice extent of an ice age) to the current interglacial state. Air warmed, ice melted, and ocean circulation was altered, but the order of these changes is still uncertain. Scientists do know that ocean circulation strongly influences the global climate. Thus, knowing what was happening in the ocean is central to understanding what drives our planet’s transition to a warmer interglacial state.
In seafloor sediment samples dating back to the time of the transition, there’s a clear and marked change in the oxygen isotopes within. Further, the change occurs in the North Atlantic earlier in the Southern Ocean, suggesting that there was a difference in the timing of the changes. Scientists recognized the potential of this information to reveal new insights about the changes in the ocean during the deglaciation.
The composition of oxygen isotopes in these seafloor samples are determined by two factors: the temperature and the isotopic composition of the surrounding seawater at the time the sediments were formed. But the hurdle that scientists have been grappling with is that it’s difficult to separate the influence of these two factors. Some scientists believe that the oxygen isotope change during the deglaciation was because melted ice was being added to the ocean, which has a much lighter isotope ratio than seawater. But other research groups suggest that it was actually ocean warming that caused the shift in the sediment samples. Zhang and his colleagues entered the arena by making use of an ocean model that could simulate both the impacts of melting ice and the changes in ocean temperature on the seafloor oxygen isotopes in order to pinpoint which was responsible.
They first tested the hypothesis that the oxygen isotopes were changing during the deglaciation because of all the freshwater being added to the ocean from the melting ice. This hypothesis is appealing because it could also explain the phasing differences between the North Atlantic and Southern Ocean. Because the North Atlantic is closer than the Southern Ocean to the Arctic (where the majority of ice melt occurs during deglaciations (Figure 3)), we’d expect to see a change in the oxygen isotopes in the North Atlantic first. But according to the model Zhang and his colleagues were using, this freshwater input would only have a mild influence on the oxygen isotopes in both the North Atlantic and Southern Ocean. Mild influences that could not come close to explaining the large changes revealed in the data. Ruling out changes in the composition of the seawater being the cause of the observed isotope changes, Zhang and the researchers next tested the theory that changing water temperature was responsible.

Testing the temperature hypothesis, Zhang and his colleagues indeed found that the North Atlantic warmed much more (about 1.4˚C) in the early phase the deglaciation, while the water temperatures in the Southern Ocean remained constant. This fit the observations that there was much more of the lighter oxygen isotope in the North Atlantic (as happens in warmer water conditions) than in the Southern Ocean at the time. Zhang and the scientists went further and formulated a mechanism explaining why the North Atlantic might have warmed more than the Southern Ocean at the start of the deglaciation.
The explanation they came up was this: during the deglaciation, the freshwater in the North Atlantic from melting ice could send cold water down from the surface to about 2,000 meter depth. During the winter season at the time, the sinking cold water would only make it to about 1,500 m, leaving the water below unperturbed and allowing it to accumulate heat. In contrast, the water at the same in the Southern Ocean didn’t seem to change much in temperature at the time due to a balance in incoming heat and incoming cold water. Thanks to Zhang and his colleagues’ work, we’ve acquired the bit of information that the North Atlantic warmed earlier than the Southern Ocean, placing another puzzle piece in the mystery of how our planet undergoes profound shifts in the state of its climate.
The huge gaps in our understanding and the scale (tens of thousands of years of our entire planet’s history) of transitions between glacial and interglacial periods only adds to the awe that such processes inspire. Zhang and his colleagues found a way to work around the constraints of seafloor oxygen isotope measurements to explain the spatially separated pattern of change in the ocean during the last deglaciation. Though so many fundamental questions about the nature of our planet’s longer-scale oscillations remain, this work brings us a step closer to understanding what exactly is happening below the sea surface when our planet undergoes the enormous transformation of switching to a new state of warmth and reduced ice cover.
Julia is a PhD student at Scripps Institution of Oceanography in La Jolla, California. Her focus is on biogeochemistry, which, as the name suggests, centers on the combined effects of biological, geological and chemical processes on the earth system. She is advised by Dr. Ralph Keeling and is modeling the global carbon cycle to better understand how much carbon dioxide ends up in the atmosphere. When not at her computer writing code, Julia can usually be found reading and/or thinking about food.

