This is a guest post by Patricia Myer. Patricia is a third-year Chemical Oceanography PhD student at the University of Connecticut. She received her BA in Environmental Science from Rensselaer Polytechnic Institute in 2017. Her current research is focused on the environmental factors affecting bioaccumulation of methylmercury into plankton.
Mercury cycles through the environment and ends up on our dinner plate in many different kinds of seafood. This is a concern because mercury is a neurotoxin which is known to have significant neurological and developmental effects. Worldwide, exposure to methylmercury comes primarily from eating marine fish, making up over 90% of mercury exposure cases in the US alone. This high rate of exposure poses significant health risks, especially for pregnant women and young children. So, how does mercury get into these fish in the first place?
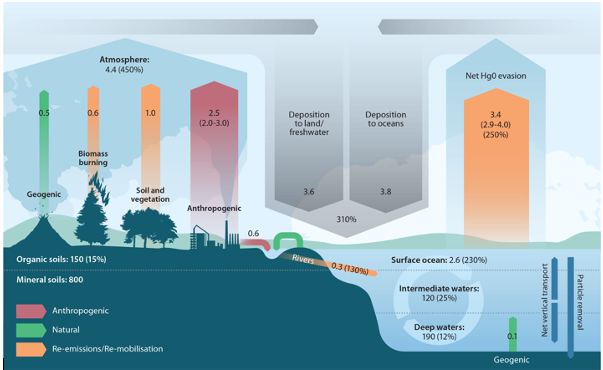
Elemental mercury (Hg0), the familiar silver liquid, is very volatile, allowing it to vaporize at temperatures far below its boiling point (similar to rubbing alcohol). Mercury vapor is released into the atmosphere by volcanic eruptions and from human sources on land (e.g. coal burning, trash burning, gold mining).
Elemental mercury does not readily react with many compounds in the atmosphere, allowing it to stay in the air. Mercury vapor can remain in the atmosphere for anywhere between several months to a year, allowing it to circle the globe. However, elemental mercury is eventually oxidized (losing electrons) by UV rays from sunlight into inorganic mercury (Hg2+), which is removed from the atmosphere in two ways: by precipitation, or by sticking onto particles in the air which settle out of the atmosphere and into the oceans. From the surface ocean, it can either be reduced (gain electrons) and become elemental mercury vapor (Hg0) once again, or it can sink into the deep ocean. As this mercury sinks, it can enter the food chain (through processes such as uptake by plankton) or make it to the seafloor, where it is buried in the sediment.
In the ocean, a number of processes can convert inorganic mercury (Hg2+) into methylmercury (CH3Hg) through a chemical reaction known as methylation. This form of mercury tends to stay in the tissues of organisms, and is taken up much faster than it can be removed (bioaccumulation). In the marine food chain, methylmercury is first taken up from the water by plankton. This mercury is then transferred to the tissues of the small fish that eat these plankton. When bigger fish eat small fish, they obtain all of the methylmercury from all of the plankton that the small fish ate.
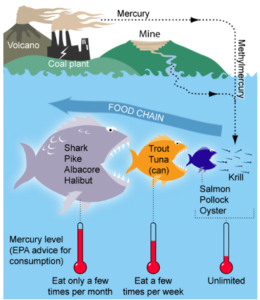
This trend continues up the food chain with a drastic increase in methylmercury at each step, resulting in high concentrations in large predatory fish at the top, in a process known as biomagnification. As humans we eat many of these predatory fish (e.g. swordfish, shark, bigeye tuna) and it is recommended to limit them in our diet due to their high mercury levels.
Next time you order seafood, make sure to avoid fish known to be higher in mercury, and opt for fish that are safer to consume such as salmon, tilapia, flounder, and many others. The FDA has a useful guide on which fish are safe to eat. This guide is aimed mainly at pregnant women and young children (due to the high risk of developmental issues from mercury exposure), but is a good resource for everyone to make informed decisions about the food we eat.
Sources
Driscoll, C. T., Mason, R. P., Chan, H. M., Jacob, D. J., & Pirrone, N. (2013). Mercury as a global pollutant: sources, pathways, and effects. Environmental science & technology, 47(10), 4967-4983. https://pubs.acs.org/doi/10.1021/es305071v
Outridge, P. M., Mason, R. P., Wang, F., Guerrero, S., & Heimbürger-Boavida, L. E. (2018). Updated global and oceanic mercury budgets for the United Nations Global Mercury Assessment 2018. Environmental science & technology, 52(20), 11466-11477. https://pubs.acs.org/doi/10.1021/acs.est.8b01246
Mason, R. P., Choi, A. L., Fitzgerald, W. F., Hammerschmidt, C. R., Lamborg, C. H., Soerensen, A. L., & Sunderland, E. M. (2012). Mercury biogeochemical cycling in the ocean and policy implications. Environmental research, 119, 101-117. https://doi.org/10.1016/j.envres.2012.03.013
I love writing of all kinds. As a PhD student at the Graduate School of Oceanography (URI), I use using genetic techniques to study phytoplankton diversity. I am interested in understanding how environmental stressors associated with climate change affect phytoplankton community dynamics and thus, overall ecosystem function. Prior to graduate school, I spent two years as a plankton analyst in the Marine Invasions Lab at the Smithsonian Environmental Research Center (SERC) studying phytoplankton in ballast water of cargo ships and gaining experience with phytoplankton taxonomy and culturing techniques. In my free time I enjoy making my own pottery and hiking in the White Mountains (NH).