Kroon, F.J., Lefèvre, C.D., Doyle, J.R. et al. 2020. DNA-based identification of predators of the corallivorous Crown-of-Thorns Starfish (Acanthastser cf. solaris) from fish faeces and gut contents. Scientific Reports, 10, 8184. https://doi.org/10.1038/s41598-020-65136-4.
Australia is having a rough 2020. In addition to an historic wildfire season and a global pandemic to worry about, Crown-of-Thorns Starfish (COTS), Acanthaster spp., are once again causing major coral loss on the Great Barrier Reef (GBR). This is far from the first time this group of invertebrate predators has wreaked havoc on this ecosystem. While periodic outbreaks are common (indeed, almost half of the decline in coral cover on the GBR between 1985 and 2012 was attributed to COTS predation), this outbreak is largely affecting areas that were spared by recent bleaching, making mitigation particularly important. Lethal injection of COTS individuals, in combination with other larger scale management techniques, attempts to not only stop the direct predation, but more importantly lessen the total amount of larvae available to recruit back to the reefs. Research has also found connections between ecosystem-level management (e.g. Marine Protected Areas) and COTS levels: reefs with fishing prohibitions have fewer COTS, and outbreaks of lesser impact. Perhaps this indicates a higher level of predation on COTS when there’s more fish? One group out of Townsville, Australia attempted to find out.

The theory: Predator Removal Hypothesis
The general idea here is not new. If you’ve taken ecology, or even just a unit during your high school biology class, you’ve probably seen a graph like this:
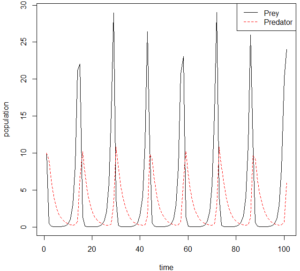
One species (in red) preys directly on another (in black). The prey species is consumed by the prey, and so decreases in numbers. This removes a food source from the predator, so they in turn decrease in number (see figure above, displaying the Lotka-Volterra model.) Without predatory pressures, the prey species’ population can explode, providing so much food to the predatory species that its population grows again on a slight time lag and the cycle repeats. If we drill down on just one spike, we can see that when predation pressure is removed for reasons natural (as above) or artificial (anthropogenic hunting or fishing pressures, habitat degradation, disease, humans removing a predator because they’re anxious that coyotes will harm their pets—you name it) the prey population spikes. Maybe you’ve seen this when deer eat your hostas, or when rabbits overrun your garden. The theory’s actually been cited as the reason for It was suggested as a potential mechanism for COTS outbreaks back in 1969, albeit referring to a species of predatory gastropod (the Giant Triton – google it, it’s 2 feet long!).[1] Research has since identified more than 80 species that prey on COTS during its different life stages, which might explain why it takes protecting the whole reef to see a difference in COTS abundance. The Australian research team had seen modeling studies testing the predator removal theory on COTS, and decided to take it one step further, examining DNA taken from the gut contents and feces of reef fishes to better understand the real world dynamics of this potential predator-prey relationship.
Experimentation: Predator Dynamics IRL
Prepping for Study:
After collecting confirmed reports from the literature of reef fish known to have consumed COTS (either from field reports, laboratory feed experiments, or gut content/fecal analyses), the team ran two pilot studies to perfect the technique detecting Pacific COTS species (A. cf. solaris) in fecal samples. In the first, detections were successful up to 7 days post-feed in two out of the five pufferfish sampled. In the second, COTS spines were found in feces of two separate species of fish. Both of these pilot studies allowed the team to perfect preservation techniques before the full study.

The Real Deal:
COTS DNA was eventually detected in 30 individuals from 18 different species of coral reef fish across eight different families, including six damselfish families. This includes several species of fish that had not previously been documented as COTS predators. One interesting question, given the fact that several of these ‘previously unknown’ predators also consume other fish species, is might that DNA have been transferred to them from a different meal rather than from direct predation on COTS (i.e. food web contamination.) The authors think not, noting that “the presence of COTS spines in some samples, combined with reported lethal predation on COTS gametes and larvae” suggest the likelihood that COTS predation is more common that previously thought.
Next Steps for COTS and Reef Management
This methodology is a step forward in COTS management: previous methods for gut content analysis is difficult as COTS remains are hard to identify and distinguish from other echinoderm remains. (This is probably why so few species had been previously identified as COTS predators.) Just as notably, the study supports the hypothesis that reef fish consume planktonic and benthic life stages of COTS on the reef, not just in the lab. Several species of fish that tested positive for COTS DNA are planktivorous feeders. In combination with the confirmed presence of COTS planktonic larvae at the reef sites where the damselfish were collected, this indicates that pelagic (meaning relating to the open waters of the sea as contrasted with waters close to structures, coasts, or bottom zones), planktonic feeding is highly probable. Damselfish are prevalent on healthy reefs. The authors suggest that damselfish, along with other planktivorous fish, could “markedly reduce the number of COTS larvae” as part of the larger goal of “lethal predation on the pelagic phase…and “predation on the settled phase” in combination with 1) reduced fisheries quotas on known COTS predators; 2) strategies to increase the abundance of fish species known to consume COTS; 3) temporal restrictions on fishing when outbreaks are predicted; and/or 4) establishing new no-take reserves at “outbreak initiation zones” and on other reefs known to have high larval connectivity. As we are always learning again and again, no species lives in a vacuum. The more we protect, the more we conserve.
[1] Endean R. Report on investigations made into aspects of the current Acanthaster planci (Crown of Thorns) infestations of certain reefs of the Great Barrier Reef. 35 (Queensland Department of Primary Industries (Fisheries Branch), Brisbane, Australia, 1969).
[1] De’ath, G., Fabricius, K., Sweatman, H. & Puotinen, M. The 27–year decline of coral cover on the Great Barrier Reef and its causes. Proc. Natl Acad. Sci. USA 109, 17995–17999 (2012).


